Structural insights into the interaction of IL-33 with its receptors
- PMID: 23980170
- PMCID: PMC3773798
- DOI: 10.1073/pnas.1308651110
Structural insights into the interaction of IL-33 with its receptors
Abstract
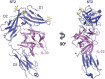
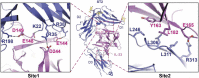
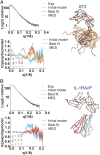
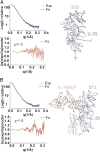
Interleukin (IL)-33 is an important member of the IL-1 family that has pleiotropic activities in innate and adaptive immune responses in host defense and disease. It signals through its ligand-binding primary receptor ST2 and IL-1 receptor accessory protein (IL-1RAcP), both of which are members of the IL-1 receptor family. To clarify the interaction of IL-33 with its receptors, we determined the crystal structure of IL-33 in complex with the ectodomain of ST2 at a resolution of 3.27 Å. Coupled with structure-based mutagenesis and binding assay, the structural results define the molecular mechanism by which ST2 specifically recognizes IL-33. Structural comparison with other ligand-receptor complexes in the IL-1 family indicates that surface-charge complementarity is critical in determining ligand-binding specificity of IL-1 primary receptors. Combined crystallography and small-angle X-ray-scattering studies reveal that ST2 possesses hinge flexibility between the D3 domain and D1D2 module, whereas IL-1RAcP exhibits a rigid conformation in the unbound state in solution. The molecular flexibility of ST2 provides structural insights into domain-level conformational change of IL-1 primary receptors upon ligand binding, and the rigidity of IL-1RAcP explains its inability to bind ligands directly. The solution architecture of IL-33-ST2-IL-1RAcP complex from small-angle X-ray-scattering analysis resembles IL-1β-IL-1RII-IL-1RAcP and IL-1β-IL-1RI-IL-1RAcP crystal structures. The collective results confer IL-33 structure-function relationships, supporting and extending a general model for ligand-receptor assembly and activation in the IL-1 family.
Keywords: SAXS; X-ray crystallography; cytokine signaling; protein–protein interaction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Liew FY, Pitman NI, McInnes IB. Disease-associated functions of IL-33: The new kid in the IL-1 family. Nat Rev Immunol. 2010;10(2):103–110. - PubMed
-
- Oboki K, Ohno T, Kajiwara N, Saito H, Nakae S. IL-33 and IL-33 receptors in host defense and diseases. Allergol Int. 2010;59(2):143–160. - PubMed
-
- Smith DE. IL-33: A tissue derived cytokine pathway involved in allergic inflammation and asthma. Clin Exp Allergy. 2010;40(2):200–208. - PubMed
-
- Palmer G, Gabay C. Interleukin-33 biology with potential insights into human diseases. Nat Rev Rheumatol. 2011;7(6):321–329. - PubMed
-
- Milovanovic M, et al. IL-33/ST2 axis in inflammation and immunopathology. Immunol Res. 2012;52(1-2):89–99. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

