Translational HIV-1 research: from routine diagnostics to new virology insights in Amsterdam, the Netherlands during 1983-2013
- PMID: 23985078
- PMCID: PMC3765835
- DOI: 10.1186/1742-4690-10-93
Translational HIV-1 research: from routine diagnostics to new virology insights in Amsterdam, the Netherlands during 1983-2013
Abstract
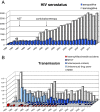
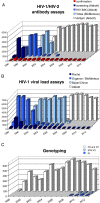
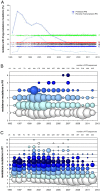
An HIV-1 diagnostic laboratory was established in the Academic Medical Center (AMC) of the University of Amsterdam after the discovery of human immunodeficiency virus (HIV) as the cause of the acquired immunodeficiency syndrome (AIDS). The first AIDS patients were diagnosed here in 1981 and since 1983 we have tested the samples of 50992 patients using a variety of assays that greatly improved over the years. We will describe some of the basic results from this diagnostic laboratory and then focus on the spin-off in terms of the development of novel virus assays to detect super-infections and ultra-sensitive assays to measure the intracellular HIV-1 RNA load. We also review several original research findings in the field of HIV-1 virology that stem from initial observations made in the diagnostic unit. This includes the study of genetic defects in the HIV-1 genome and time trends of the replication fitness over 30 years of viral evolution, but also the description of novel HIV-1 variants in difficult-to-diagnose clinical specimen.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

