Postnatal refinement of auditory hair cell planar polarity deficits occurs in the absence of Vangl2
- PMID: 23986237
- PMCID: PMC3756750
- DOI: 10.1523/JNEUROSCI.1307-13.2013
Postnatal refinement of auditory hair cell planar polarity deficits occurs in the absence of Vangl2
Abstract
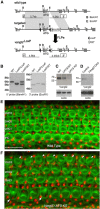
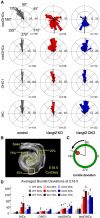
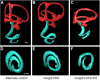
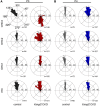
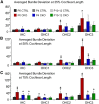
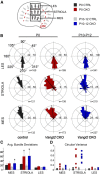
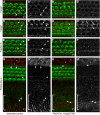
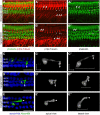
The distinctive planar polarity of auditory hair cells is evident in the polarized organization of the stereociliary bundle. Mutations in the core planar cell polarity gene Van Gogh-like 2 (Vangl2) result in hair cells that fail to properly orient their stereociliary bundles along the mediolateral axis of the cochlea. The severity of this phenotype is graded along the length of the cochlea, similar to the hair cell differentiation gradient, suggesting that an active refinement process corrects planar polarity phenotypes in Vangl2 knock-out (KO) mice. Because Vangl2 gene deletions are lethal, Vangl2 conditional knock-outs (CKOs) were generated to test this hypothesis. When crossed with Pax2-Cre, Vangl2 is deleted from the inner ear, yielding planar polarity phenotypes similar to Vangl2 KOs at late embryonic stages except that Vangl2 CKO mice are viable and do not have craniorachischisis like Vangl2 KOs. Quantification of planar polarity deficits through postnatal development demonstrates the activity of a Vangl2-independent refinement process that rescues the planar polarity phenotype within 10 d of birth. In contrast, the Pax2-Cre;Vangl2 CKO has profound changes in the shape and distribution of outer pillar cell and Deiters' cell phalangeal processes that are not corrected during the period of planar polarity refinement. Auditory brainstem response analyses of adult mice show a 10-15 dB shift in auditory threshold, and distortion product otoacoustic emission measurements indicate that this mild hearing deficit is of cochlear origin. Together, these data demonstrate a Vangl2-independent refinement mechanism that actively reorients auditory stereociliary bundles and reveals an unexpected role of Vangl2 during supporting cell morphogenesis.
Figures



References
-
- Andre P, Wang Q, Wang N, Gao B, Schilit A, Halford MM, Stacker SA, Zhang X, Yang Y. The Wnt coreceptor Ryk regulates Wnt/planar cell polarity by modulating the degradation of the core planar cell polarity component Vangl2. J Biol Chem. 2012;287:44518–44525. doi: 10.1074/jbc.M112.414441. - DOI - PMC - PubMed
-
- Collado MS, Thiede BR, Baker W, Askew C, Igbani LM, Corwin JT. The postnatal accumulation of junctional E-cadherin is inversely correlated with the capacity for supporting cells to convert directly into sensory hair cells in mammalian balance organs. J Neurosci. 2011;31:11855–11866. doi: 10.1523/JNEUROSCI.2525-11.2011. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
