Integration of biosynthesis and long-distance transport establish organ-specific glucosinolate profiles in vegetative Arabidopsis
- PMID: 23995084
- PMCID: PMC3784604
- DOI: 10.1105/tpc.113.110890
Integration of biosynthesis and long-distance transport establish organ-specific glucosinolate profiles in vegetative Arabidopsis
Abstract
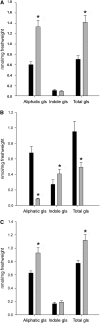
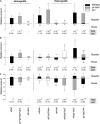
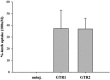
Although it is essential for plant survival to synthesize and transport defense compounds, little is known about the coordination of these processes. Here, we investigate the above- and belowground source-sink relationship of the defense compounds glucosinolates in vegetative Arabidopsis thaliana. In vivo feeding experiments demonstrate that the glucosinolate transporters1 and 2 (GTR1 and GTR2), which are essential for accumulation of glucosinolates in seeds, are likely to also be involved in bidirectional distribution of glucosinolates between the roots and rosettes, indicating phloem and xylem as their transport pathways. Grafting of wild-type, biosynthetic, and transport mutants show that both the rosette and roots are able to synthesize aliphatic and indole glucosinolates. While rosettes constitute the major source and storage site for short-chained aliphatic glucosinolates, long-chained aliphatic glucosinolates are synthesized both in roots and rosettes with roots as the major storage site. Our grafting experiments thus indicate that in vegetative Arabidopsis, GTR1 and GTR2 are involved in bidirectional long-distance transport of aliphatic but not indole glucosinolates. Our data further suggest that the distinct rosette and root glucosinolate profiles in Arabidopsis are shaped by long-distance transport and spatially separated biosynthesis, suggesting that integration of these processes is critical for plant fitness in complex natural environments.
Figures



References
-
- Brady S.M., Orlando D.A., Lee J.Y., Wang J.Y., Koch J., Dinneny J.R., Mace D., Ohler U., Benfey P.N. (2007). A high-resolution root spatiotemporal map reveals dominant expression patterns. Science 318: 801–806 - PubMed
-
- Brown P.D., Tokuhisa J.G., Reichelt M., Gershenzon J. (2003). Variation of glucosinolate accumulation among different organs and developmental stages of Arabidopsis thaliana. Phytochemistry 62: 471–481 - PubMed
-
- Brudenell A.J.P., Griffiths H., Rossiter J.T., Baker D.A. (1999). The phloem mobility of glucosinolates. J. Exp. Bot. 50: 745–756
-
- Chen, S., Glawischnig, E., Jorgensen, K., Naur, P., Jorgensen, B., Olsen, C.E., Hansen, C.H., Rasmussen, H., Pickett, J.A., and Halkier, B.A. (2003). CYP79F1 and CYP79F2 have distinct functions in the biosynthesis of aliphatic glucosinolates in Arabidopsis Plant J. 33: 923–937. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

