Efficacy and safety outcomes of oral anticoagulants and antiplatelet drugs in the secondary prevention of venous thromboembolism: systematic review and network meta-analysis
- PMID: 23996149
- PMCID: PMC3758108
- DOI: 10.1136/bmj.f5133
Efficacy and safety outcomes of oral anticoagulants and antiplatelet drugs in the secondary prevention of venous thromboembolism: systematic review and network meta-analysis
Abstract
Objective: To summarise and compare the efficacy and safety of various oral anticoagulants (dabigatran, rivaroxaban, apixaban, and vitamin K antagonists) and antiplatelet agents (acetylsalicylic acid) for the secondary prevention of venous thromboembolism.
Design: Systematic review and network meta-analysis.
Data sources: Literature search using Medline (1950 to present), Embase (1980 to present), and the Cochrane Register of Controlled Trials using the OVID interface. Publications from potentially relevant journals were also searched by hand.
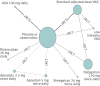
Review methods: Randomised controlled trials of patients receiving anticoagulants, antiplatelet drugs, or placebo or observation for secondary prevention of venous thromboembolism. Selected outcomes were rates of recurrent venous thromboembolism and major bleeding. Two reviewers independently extracted data onto standardised forms.
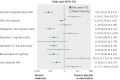
Results: 12 articles met our inclusion criteria, with 11,999 patients evaluated for efficacy and 12,167 for safety. All treatments reduced the risk of recurrent venous thromboembolism. Compared with placebo or observation, vitamin K antagonists at a standard adjusted dose (target international normalised ratio 2.0-3.0) showed the highest risk difference (odds ratio 0.07; 95% credible interval 0.03 to 0.15) and acetylsalicylic acid showed the lowest risk difference (0.65; 0.39 to 1.03). Risk of major bleeding was higher with a standard adjusted dose of vitamin K antagonists (5.24; 1.78 to 18.25) than with placebo or observation. Fatal recurrent venous thromboembolism and fatal bleeding were rare. Detailed subgroup and individual patient level data were not available.
Conclusions: All oral anticoagulants and antiplatelet agents investigated in this analysis were associated with a reduced recurrence of venous thromboembolism compared with placebo or observation, although acetylsalicylic acid was associated with the lowest risk reduction. Vitamin K antagonists given at a standard adjusted dose was associated with the greatest risk reduction in recurrent venous thromboembolism, but also the greatest risk of major bleeding.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at
Figures

Comment in
-
Secondary prevention of venous thromboembolism.BMJ. 2013 Sep 9;347:f5440. doi: 10.1136/bmj.f5440. BMJ. 2013. PMID: 24018259 No abstract available.
References
-
- Cohen AT, Agnelli G, Anderson FA, Arcelus JI, Bergqvist D, Brecht JG, et al. Venous thromboembolism (VTE) in Europe. The number of VTE events and associated morbidity and mortality. Thromb Haemost 2007;98:756-64. - PubMed
-
- Ageno W, Prandoni P, Romualdi E, Ghirarduzzi A, Dentali F, Pesavento R, et al. The metabolic syndrome and the risk of venous thrombosis: a case-control study. J Thromb Haemost 2006;4:1914-8. - PubMed
-
- Heit JA. The epidemiology of venous thromboembolism in the community: implications for prevention and management. J Thromb Thrombolysis 2006;21:23-9. - PubMed
-
- White RH. The epidemiology of venous thromboembolism. Circulation 2003;107:I4-8. - PubMed
-
- Prins MH, Marchiori A. Risk of recurrent venous thomboembolism—expanding the frontier. Thromb Haemost 2002;87:1-3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
