Effects of prostaglandin F(2α) on adipocyte biology relevant to graves' orbitopathy
- PMID: 24001049
- PMCID: PMC3868384
- DOI: 10.1089/thy.2013.0194
Effects of prostaglandin F(2α) on adipocyte biology relevant to graves' orbitopathy
Abstract
Background: In Graves' orbitopathy (GO), increased proliferation, excess adipogenesis, and hyaluronan overproduction produce GO exophthalmos. Enophthalmos occurs in some glaucoma patients treated with Bimatoprost (prostaglandin F2α, PGF2α) eye drops. We hypothesized that enophthalmos is secondary to reductions in orbital tissue proliferation, adipogenesis, and/or increased lipolysis. We aimed to determine which of these is affected by PGF2α by using the 3T3-L1 murine preadipocyte cell line and primary human orbital fibroblasts (OFs) from GO patients (n=5) and non-GO (n=5).
Methods: 3T3-L1 cells and orbital OFs were cultured alone or with PGF2α (all experiments used 10(-8) to 10(-6) M) and counted on days 1/2/3 or 5, respectively; cell cycle analysis (flow cytometry) was applied. Adipogenesis (in the presence/absence of PGF2α) was evaluated (day 7 or 15 for 3T3-L1 and primary cells, respectively) morphologically by Oil Red O staining and quantitative polymerase chain reaction measurement of adipogenesis markers (glycerol-3-phosphate dehydrogenase and lipoprotein lipase, respectively). For lipolysis, in vitro-differentiated 3T3-L1 or mature orbital adipocytes were incubated with norepinephrine and PGF2α and free glycerol was assayed. Appropriate statistical tests were applied.
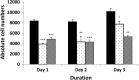
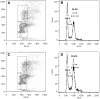
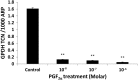
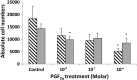
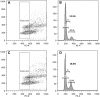
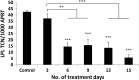
Results: The population doubling time of 3T3-L1 was 27.3±1.4 hours-significantly increased by dimethyl sulfoxide 0.02% to 44.6±4.8 hours (p=0.007) and further significantly increased (p=0.049 compared with dimethyl sulfoxide) by 10(-8) M PGF2α to 93.6±19.0 hours, indicating reduced proliferation, which was caused by prolongation of G2/M. GO OFs proliferated significantly more rapidly than non-GO (population doubling time 5.36±0.34 or 6.63±0.35 days, respectively, p=0.035), but the proliferation of both was significantly reduced (dose dependent from 10(-8) M) by PGF2α, again with prolongation of G2/M. Adipogenesis in 3T3-L1 cells was minimally affected by PGF2α when assessed morphologically, but the drug significantly reduced transcripts of the glycerol-3-phosphate dehydrogenase differentiation marker. GO OFs displayed significantly higher adipogenic potential than non-GO, but in both populations, adipogenesis, evaluated by all 3 methods, was significantly reduced (dose dependent from 10(-8) M) by PGF2α. There was no effect of PGF2α on basal or norepinephrine-induced lipolysis, in 3T3-L1 or human OFs, either GO or non-GO.
Conclusions: The results demonstrate that PGF2α significantly reduces proliferation and adipogenesis and that human OFs are more sensitive to its effects than 3T3-L1. Consequently, PGF2α could be effective in the treatment of GO.
Figures

References
-
- Eisenberg DL, Toris CB, Camras CB.2002Bimatoprost and travoprost: a review of recent studies of two new glaucoma drugs. Surv Ophthalmol 47Suppl 1:S105–S115 - PubMed
-
- Peplinski LS, Albiani Smith K.2004Deepening of lid sulcus from topical bimatoprost therapy. Optom Vis Sci 81:574–577 - PubMed
-
- Filippopoulos T, Paula JS, Torun N, Hatton MP, Pasquale LR, Grosskreutz CL.2008Periorbital changes associated with topical bimatoprost. Ophthal Plast Reconstr Surg 24:302–307 - PubMed
-
- Tappeiner C, Perren B, Iliev ME, Frueh BE, Goldblum D.2008[Orbital fat atrophy in glaucoma patients treated with topical bimatoprost—can bimatoprost cause enophthalmos?]. Klin Monbl Augenheilkd 225:443–445 - PubMed
-
- Yam JC, Yuen NS, Chan CW.2009Bilateral deepening of upper lid sulcus from topical bimatoprost therapy. J Ocul Pharmacol Ther 25:471–472 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

