Sequestration and histopathology in Plasmodium chabaudi malaria are influenced by the immune response in an organ-specific manner
- PMID: 24003897
- PMCID: PMC4234010
- DOI: 10.1111/cmi.12212
Sequestration and histopathology in Plasmodium chabaudi malaria are influenced by the immune response in an organ-specific manner
Abstract
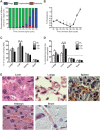
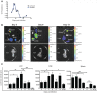
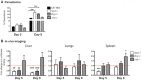
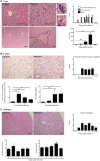
Infection with the malaria parasite, Plasmodium, is associated with a strong inflammatory response and parasite cytoadhesion (sequestration) in several organs. Here, we have carried out a systematic study of sequestration and histopathology during infection of C57Bl/6 mice with Plasmodium chabaudi AS and determined the influence of the immune response. This parasite sequesters predominantly in liver and lung, but not in the brain, kidney or gut. Histopathological changes occur in multiple organs during the acute infection, but are not restricted to the organs where sequestration takes place. Adaptive immunity, and signalling through the IFNγ receptor increased sequestration and histopathology in the liver, but not in the lung, suggesting that there are differences in the adhesion molecules and/or parasite ligands utilized and mechanisms of pathogenesis in these two organs. Exacerbation of pro-inflammatory responses during infection by deletion of the il10 gene resultsin the aggravation of damage to lung and kidney irrespective of the degree of sequestration. The immune response therefore affected both sequestration and histopathology in an organ-specific manner. P. chabaudi AS provides a good model to investigate the influence of the host response on the sequestration and specific organ pathology, which is applicable to human malaria.
© 2013 Crown copyright. Cellular Microbiology © 2013 John Wiley & Sons Ltd. This article is published with the permission of the Controller of HMSO and the Queen's Printer for Scotland.
Figures

Similar articles
-
ICAM-1 is a key receptor mediating cytoadherence and pathology in the Plasmodium chabaudi malaria model.Malar J. 2017 May 3;16(1):185. doi: 10.1186/s12936-017-1834-8. Malar J. 2017. PMID: 28468674 Free PMC article.
-
Plasmodium chabaudi-infected erythrocytes adhere to CD36 and bind to microvascular endothelial cells in an organ-specific way.Infect Immun. 2000 Jul;68(7):4135-44. doi: 10.1128/IAI.68.7.4135-4144.2000. Infect Immun. 2000. PMID: 10858230 Free PMC article.
-
Dissemination of non-typhoidal Salmonella during Plasmodium chabaudi infection affects anti-malarial immunity.Parasitol Res. 2019 Jul;118(7):2277-2285. doi: 10.1007/s00436-019-06349-z. Epub 2019 May 23. Parasitol Res. 2019. PMID: 31119381 Free PMC article.
-
The contribution of Plasmodium chabaudi to our understanding of malaria.Trends Parasitol. 2012 Feb;28(2):73-82. doi: 10.1016/j.pt.2011.10.006. Epub 2011 Nov 17. Trends Parasitol. 2012. PMID: 22100995 Free PMC article. Review.
-
A dual role for B cells in Plasmodium chabaudi chabaudi (AS) infection?Res Immunol. 1994 Jul-Aug;145(6):412-9. doi: 10.1016/s0923-2494(94)80170-3. Res Immunol. 1994. PMID: 7899705 Review.
Cited by
-
Malaria in pregnancy: the relevance of animal models for vaccine development.Lab Anim (NY). 2017 Oct 6;46(10):388-398. doi: 10.1038/laban.1349. Lab Anim (NY). 2017. PMID: 28984865 Free PMC article. Review.
-
Signatures of malaria-associated pathology revealed by high-resolution whole-blood transcriptomics in a rodent model of malaria.Sci Rep. 2017 Feb 3;7:41722. doi: 10.1038/srep41722. Sci Rep. 2017. PMID: 28155887 Free PMC article.
-
Gene expression of the liver of vaccination-protected mice in response to early patent infections of Plasmodium chabaudi blood-stage malaria.Malar J. 2018 May 29;17(1):215. doi: 10.1186/s12936-018-2366-6. Malar J. 2018. PMID: 29843710 Free PMC article.
-
Inducible mechanisms of disease tolerance provide an alternative strategy of acquired immunity to malaria.Elife. 2021 Mar 23;10:e63838. doi: 10.7554/eLife.63838. Elife. 2021. PMID: 33752799 Free PMC article.
-
Mouse Models for Unravelling Immunology of Blood Stage Malaria.Vaccines (Basel). 2022 Sep 14;10(9):1525. doi: 10.3390/vaccines10091525. Vaccines (Basel). 2022. PMID: 36146602 Free PMC article. Review.
References
-
- Aird WC. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ Res. 100:158–173. - PubMed
-
- Aird WC. Phenotypic heterogeneity of the endothelium: II. Representative vascular beds. Circ Res. 100:174–190. 2007a–2007b. - PubMed
-
- Amante FH, Haque A, Stanley AC, Rivera Fde L, Randall LM, Wilson YA, et al. Immune-mediated mechanisms of parasite tissue sequestration during experimental cerebral malaria. J Immunol. 2010;185:3632–3642. - PubMed
-
- Anstey NM, Russell B, Yeo TW, Price RN. The pathophysiology of vivax malaria. Trends Parasitol. 2009;25:220–227. , and. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

