Endosomal acidification by Na+/H+ exchanger NHE5 regulates TrkA cell-surface targeting and NGF-induced PI3K signaling
- PMID: 24006492
- PMCID: PMC3814139
- DOI: 10.1091/mbc.E12-06-0445
Endosomal acidification by Na+/H+ exchanger NHE5 regulates TrkA cell-surface targeting and NGF-induced PI3K signaling
Abstract
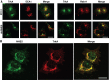
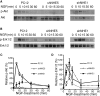
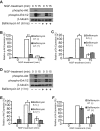
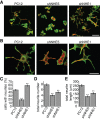
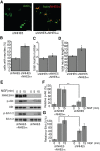
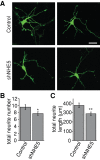
To facilitate polarized vesicular trafficking and signal transduction, neuronal endosomes have evolved sophisticated mechanisms for pH homeostasis. NHE5 is a member of the Na(+)/H(+) exchanger family and is abundantly expressed in neurons and associates with recycling endosomes. Here we show that NHE5 potently acidifies recycling endosomes in PC12 cells. NHE5 depletion by plasmid-based short hairpin RNA significantly reduces cell surface abundance of TrkA, an effect similar to that observed after treatment with the V-ATPase inhibitor bafilomycin. A series of cell-surface biotinylation experiments suggests that anterograde trafficking of TrkA from recycling endosomes to plasma membrane is the likeliest target affected by NHE5 depletion. NHE5 knockdown reduces phosphorylation of Akt and Erk1/2 and impairs neurite outgrowth in response to nerve growth factor (NGF) treatment. Of interest, although both phosphoinositide 3-kinase-Akt and Erk signaling are activated by NGF-TrkA, NGF-induced Akt-phosphorylation appears to be more sensitively affected by perturbed endosomal pH. Furthermore, NHE5 depletion in rat cortical neurons in primary culture also inhibits neurite formation. These results collectively suggest that endosomal pH modulates trafficking of Trk-family receptor tyrosine kinases, neurotrophin signaling, and possibly neuronal differentiation.
Figures




References
-
- Arimura N, et al. Anterograde transport of TrkB in axons is mediated by direct interaction with Slp1 and Rab27. Dev Cell. 2009;16:675–686. - PubMed
-
- Arimura N, Kaibuchi K. Neuronal polarity: from extracellular signals to intracellular mechanisms. Nat Rev Neurosci. 2007;8:194–205. - PubMed
-
- Attaphitaya S, Park K, Melvin JE. Molecular cloning and functional expression of a rat Na+/H+ exchanger (NHE5) highly expressed in brain. J Biol Chem. 1999;274:4383–4388. - PubMed
-
- Baass PC, Di Guglielmo GM, Authier F, Posner BI, Bergeron JJ. Compartmentalized signal transduction by receptor tyrosine kinases. Trends Cell Biol. 1995;5:465–470. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

