Activated microglia mediate synapse loss and short-term memory deficits in a mouse model of transthyretin-related oculoleptomeningeal amyloidosis
- PMID: 24008733
- PMCID: PMC3789183
- DOI: 10.1038/cddis.2013.325
Activated microglia mediate synapse loss and short-term memory deficits in a mouse model of transthyretin-related oculoleptomeningeal amyloidosis
Abstract
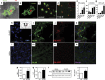
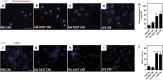
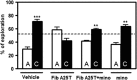
Oculoleptomeningeal amyloidosis (OA) is a fatal and untreatable hereditary disease characterized by the accumulation of transthyretin (TTR) amyloid within the central nervous system. The mechanisms underlying the pathogenesis of OA, and in particular how amyloid triggers neuronal damage, are still unknown. Here, we show that amyloid fibrils formed by a mutant form of TTR, A25T, activate microglia, leading to the secretion of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and nitric oxide. Further, we found that A25T amyloid fibrils induce the activation of Akt, culminating in the translocation of NFκB to the nucleus of microglia. While A25T fibrils were not directly toxic to neurons, the exposure of neuronal cultures to media conditioned by fibril-activated microglia caused synapse loss that culminated in extensive neuronal death via apoptosis. Finally, intracerebroventricular (i.c.v.) injection of A25T fibrils caused microgliosis, increased brain TNF-α and IL-6 levels and cognitive deficits in mice, which could be prevented by minocycline treatment. These results indicate that A25T fibrils act as pro-inflammatory agents in OA, activating microglia and causing neuronal damage.
Figures



Similar articles
-
Energetic characteristics of the new transthyretin variant A25T may explain its atypical central nervous system pathology.Lab Invest. 2003 Mar;83(3):409-17. doi: 10.1097/01.lab.0000059937.11023.1f. Lab Invest. 2003. PMID: 12649341
-
Neoechinulin A suppresses amyloid-β oligomer-induced microglia activation and thereby protects PC-12 cells from inflammation-mediated toxicity.Neurotoxicology. 2013 Mar;35:30-40. doi: 10.1016/j.neuro.2012.12.004. Epub 2012 Dec 21. Neurotoxicology. 2013. PMID: 23261590
-
α-Iso-cubebenol inhibits inflammation-mediated neurotoxicity and amyloid beta 1-42 fibril-induced microglial activation.J Pharm Pharmacol. 2014 Jan;66(1):93-105. doi: 10.1111/jphp.12160. Epub 2013 Oct 21. J Pharm Pharmacol. 2014. PMID: 24138316
-
Familial amyloidotic polyneuropathy: protein aggregation in the peripheral nervous system.J Mol Neurosci. 2004;23(1-2):35-40. doi: 10.1385/jmn:23:1-2:035. J Mol Neurosci. 2004. PMID: 15126690 Review.
-
[Cerebral amyloid angiopathy with familial transthyretin-derived oculoleptomeningeal amyloidosis].Brain Nerve. 2013 Jul;65(7):831-42. Brain Nerve. 2013. PMID: 23832986 Review. Japanese.
Cited by
-
Minocycline causes widespread cell death and increases microglial labeling in the neonatal mouse brain.Dev Neurobiol. 2017 Jun;77(6):753-766. doi: 10.1002/dneu.22457. Epub 2016 Oct 14. Dev Neurobiol. 2017. PMID: 27706925 Free PMC article.
-
Glial Contribution to Excitatory and Inhibitory Synapse Loss in Neurodegeneration.Front Cell Neurosci. 2019 Feb 26;13:63. doi: 10.3389/fncel.2019.00063. eCollection 2019. Front Cell Neurosci. 2019. PMID: 30863284 Free PMC article. Review.
-
A novel combinational approach of microstimulation and bioluminescence imaging to study the mechanisms of action of cerebral electrical stimulation in mice.J Physiol. 2015 May 15;593(10):2257-78. doi: 10.1113/jphysiol.2014.287243. Epub 2015 Mar 24. J Physiol. 2015. PMID: 25653107 Free PMC article.
-
Amyloid Proteins and Peripheral Neuropathy.Cells. 2020 Jun 26;9(6):1553. doi: 10.3390/cells9061553. Cells. 2020. PMID: 32604774 Free PMC article. Review.
-
HIV Neuroinfection and Alzheimer's Disease: Similarities and Potential Links?Front Cell Neurosci. 2018 Sep 11;12:307. doi: 10.3389/fncel.2018.00307. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30254568 Free PMC article.
References
-
- Shimizu Y, Takeuchi M, Matsumura M, Tokuda T, Iwata M. A case of biopsy-proven leptomeningeal amyloidosis and intravenous Ig-responsive polyneuropathy associated with the Ala25Thr transthyretin gene mutation. Amyloid. 2006;13:37–41. - PubMed
-
- Hagiwara K, Ochi H, Suzuki S, Shimizu Y, Tokuda T, Murai H, et al. Highly selective leptomeningeal amyloidosis with transthyretin variant Ala25Thr. Neurology. 2009;72:1358–1360. - PubMed
-
- De Carolis P, Galeotti M, Ficarra G, Masetti M, Grimaldi D, Cortelli P. Fatal cerebral haemorrhage after liver transplantation in a patient with transthyretin variant (gly53glu) amyloidosis. Neurol Sci. 2006;27:352–354. - PubMed
-
- Rinaldi L, Busani S, Pasetto A, Girardis M. Posttransplant cerebral hemorrhage in a patient with familial amyloidotic polyneuropathy. Transplantation. 2006;81:134–135. - PubMed
-
- Owa M, Takei Y, Hashikura Y, Kawasaki S, Koyama M, Ikeda S. Recurrent cerebral embolism in a familial amyloid polyneuropathy patient who received partial liver transplantation from a living donor. Intern Med. 2001;40:259–264. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

