Moving domain computational fluid dynamics to interface with an embryonic model of cardiac morphogenesis
- PMID: 24009714
- PMCID: PMC3751826
- DOI: 10.1371/journal.pone.0072924
Moving domain computational fluid dynamics to interface with an embryonic model of cardiac morphogenesis
Abstract
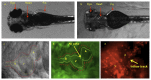
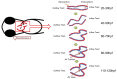
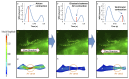
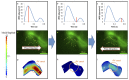
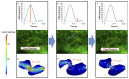
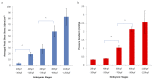
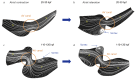
Peristaltic contraction of the embryonic heart tube produces time- and spatial-varying wall shear stress (WSS) and pressure gradients (∇P) across the atrioventricular (AV) canal. Zebrafish (Danio rerio) are a genetically tractable system to investigate cardiac morphogenesis. The use of Tg(fli1a:EGFP) (y1) transgenic embryos allowed for delineation and two-dimensional reconstruction of the endocardium. This time-varying wall motion was then prescribed in a two-dimensional moving domain computational fluid dynamics (CFD) model, providing new insights into spatial and temporal variations in WSS and ∇P during cardiac development. The CFD simulations were validated with particle image velocimetry (PIV) across the atrioventricular (AV) canal, revealing an increase in both velocities and heart rates, but a decrease in the duration of atrial systole from early to later stages. At 20-30 hours post fertilization (hpf), simulation results revealed bidirectional WSS across the AV canal in the heart tube in response to peristaltic motion of the wall. At 40-50 hpf, the tube structure undergoes cardiac looping, accompanied by a nearly 3-fold increase in WSS magnitude. At 110-120 hpf, distinct AV valve, atrium, ventricle, and bulbus arteriosus form, accompanied by incremental increases in both WSS magnitude and ∇P, but a decrease in bi-directional flow. Laminar flow develops across the AV canal at 20-30 hpf, and persists at 110-120 hpf. Reynolds numbers at the AV canal increase from 0.07±0.03 at 20-30 hpf to 0.23±0.07 at 110-120 hpf (p< 0.05, n=6), whereas Womersley numbers remain relatively unchanged from 0.11 to 0.13. Our moving domain simulations highlights hemodynamic changes in relation to cardiac morphogenesis; thereby, providing a 2-D quantitative approach to complement imaging analysis.
Conflict of interest statement
Figures

References
-
- Hove JR, Köster RW, Forouhar AS, Acevedo-Bolton G, Fraser SE et al. (2003) Intracardiac fluid forces are an essential epigenetic factor for embryonic cardiogenesis. Nature 421: 172-177. doi:10.1038/nature01282. PubMed: 12520305. - DOI - PubMed
-
- Forouhar AS, Liebling M, Hickerson A, Nasiraei-Moghaddam A, Tsai HJ et al. (2006) The embryonic vertebrate heart tube is a dynamic suction pump. Science 312: 751-753. doi:10.1126/science.1123775. PubMed: 16675702. - DOI - PubMed
-
- Scherz PJ, Huisken J, Sahai-Hernandez P, Stainier DY (2008) High-speed imaging of developing heart valves reveals interplay of morphogenesis and function. Development 135: 1179-1187. doi:10.1242/dev.010694. PubMed: 18272595. - DOI - PubMed
-
- Grego-Bessa J, Luna-Zurita L, del Monte G, Bolós V, Melgar P et al. (2007) Notch signaling is essential for ventricular chamber development. Dev Cell 12: 415-429. doi:10.1016/j.devcel.2006.12.011. PubMed: 17336907. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

