Helicobacter bilis gamma-glutamyltranspeptidase enhances inflammatory stress response via oxidative stress in colon epithelial cells
- PMID: 24009737
- PMCID: PMC3751837
- DOI: 10.1371/journal.pone.0073160
Helicobacter bilis gamma-glutamyltranspeptidase enhances inflammatory stress response via oxidative stress in colon epithelial cells
Abstract
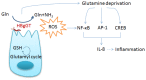
Helicobacter bilis (H. bilis) infection is associated with cases of inflammatory bowel Disease, thyphlocolitis, hepatitis and cholecystitis. However, little is known about the bacterial virulence determinants or the molecular mechanisms involved. Recently, H. bilis γ-glutamyltranspeptidase (HBgGT) was shown to be a virulence factor decreasing host cell viability. Bacterial gGTs play a key role in synthesis and degradation of glutathione and enables the bacteria to utilize extracellular glutamine and glutathione as sources of glutamate. gGT-mediated loss of cell viability has so far been linked to DNA damage via oxidative stress, but the signaling cascades involved herein have not been described. In this study, we identified enhanced ROS production induced by HBgGT as a central factor involved in the activation of the oxidative stress response cascades, which finally activate CREB, AP-1 and NF-κB in H. bilis infected colon cancer cells. IL-8, an important pro-inflammatory chemokine that is a common downstream target of these transcription factors, was up-regulated upon H. bilis infection in an HBgGT dependent manner. Moreover, the induction of these signaling responses and inflammatory cytokine production in host cells could be linked to HBgGT-mediated glutamine deprivation. This study implicates for the first time HBgGT as an important regulator of signaling cascades regulating inflammation in H. bilis infected host epithelial cells that could be responsible for induction of inflammatory disorders by the bacterium.
Conflict of interest statement
Figures


Similar articles
-
Helicobacter bilis infection induces oxidative stress in and enhances the proliferation of human cholangiocytes.Helicobacter. 2022 Aug;27(4):e12908. doi: 10.1111/hel.12908. Epub 2022 Jun 6. Helicobacter. 2022. PMID: 35661483
-
Evidence for conserved function of γ-glutamyltranspeptidase in Helicobacter genus.PLoS One. 2012;7(2):e30543. doi: 10.1371/journal.pone.0030543. Epub 2012 Feb 14. PLoS One. 2012. PMID: 22348013 Free PMC article.
-
Gamma-glutamyltranspeptidase expression by Helicobacter saguini, an enterohepatic Helicobacter species isolated from cotton top tamarins with chronic colitis.Cell Microbiol. 2019 Mar;21(3):e12968. doi: 10.1111/cmi.12968. Epub 2018 Nov 14. Cell Microbiol. 2019. PMID: 30365223 Free PMC article.
-
Oxidant stress regulation of IL-8 and ICAM-1 gene expression: differential activation and binding of the transcription factors AP-1 and NF-kappaB (Review).Int J Mol Med. 1999 Sep;4(3):223-30. doi: 10.3892/ijmm.4.3.223. Int J Mol Med. 1999. PMID: 10425270 Review.
-
Chemokine expression in Helicobacter pylori-infected gastric mucosa.J Gastroenterol. 1998 Oct;33(5):613-7. doi: 10.1007/s005350050146. J Gastroenterol. 1998. PMID: 9773923 Review.
Cited by
-
Role of Campylobacter jejuni gamma-glutamyl transpeptidase on epithelial cell apoptosis and lymphocyte proliferation.Gut Pathog. 2014 Jun 12;6:20. doi: 10.1186/1757-4749-6-20. eCollection 2014. Gut Pathog. 2014. PMID: 24995041 Free PMC article.
-
The complex amino acid diet of Francisella in infected macrophages.Front Cell Infect Microbiol. 2015 Feb 6;5:9. doi: 10.3389/fcimb.2015.00009. eCollection 2015. Front Cell Infect Microbiol. 2015. PMID: 25705612 Free PMC article. Review.
-
Association between Helicobacter spp. infections and hepatobiliary malignancies: a review.World J Gastroenterol. 2015 Feb 7;21(5):1414-23. doi: 10.3748/wjg.v21.i5.1414. World J Gastroenterol. 2015. PMID: 25663761 Free PMC article. Review.
-
Helicobacter saguini, a Novel Helicobacter Isolated from Cotton-Top Tamarins with Ulcerative Colitis, Has Proinflammatory Properties and Induces Typhlocolitis and Dysplasia in Gnotobiotic IL-10-/- Mice.Infect Immun. 2016 Jul 21;84(8):2307-2316. doi: 10.1128/IAI.00235-16. Print 2016 Aug. Infect Immun. 2016. PMID: 27245408 Free PMC article.
-
High Levels of Gamma-Glutamyl Transferase and Indocyanine Green Retention Rate at 15 min as Preoperative Predictors of Tumor Recurrence in Patients With Hepatocellular Carcinoma.Medicine (Baltimore). 2015 May;94(21):e810. doi: 10.1097/MD.0000000000000810. Medicine (Baltimore). 2015. PMID: 26020384 Free PMC article.
References
-
- Fox JG (2007) Helicobacter bilis: bacterial provocateur orchestrates host immune responses to commensal flora in a model of inflammatory bowel disease. Gut 56: 898-900. doi:10.1136/gut.2006.115428. PubMed: 17566023. - DOI - PMC - PubMed
-
- Rossi M, Zanoni RG, Hänninen ML (2010) Delineation of two Helicobacter bilis genomospecies: implications for systematics and evolution. Int J Syst Evol Microbiol 60: 2392-2397. doi:10.1099/ijs.0.016287-0. PubMed: 19946059. - DOI - PubMed
-
- Jergens AE, Wilson-Welder JH, Dorn A, Henderson A, Liu Z et al. (2007) Helicobacter bilis triggers persistent immune reactivity to antigens derived from the commensal bacteria in gnotobiotic C3H/HeN mice. Gut 56: 934-940. doi:10.1136/gut.2006.099242. PubMed: 17145736. - DOI - PMC - PubMed
-
- Liu Z, Ramer-Tait AE, Henderson AL, Demirkale CY, Nettleton D et al. (2011) Helicobacter bilis colonization enhances susceptibility to Typhlocolitis following an inflammatory trigger. Dig Dis Sci 56: 2838-2848. doi:10.1007/s10620-011-1701-3. PubMed: 21503679. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

