Environmental enrichment rescues binocular matching of orientation preference in mice that have a precocious critical period
- PMID: 24012279
- PMCID: PMC3796011
- DOI: 10.1016/j.neuron.2013.07.023
Environmental enrichment rescues binocular matching of orientation preference in mice that have a precocious critical period
Abstract
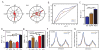
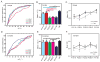
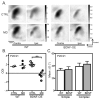
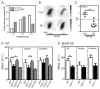
Experience shapes neural circuits during critical periods in early life. The timing of critical periods is regulated by both genetics and the environment. Here we study the functional significance of such temporal regulations in the mouse primary visual cortex, where critical period plasticity drives binocular matching of orientation preference. We find that the binocular matching is permanently disrupted in mice that have a precocious critical period due to genetically enhanced inhibition. The disruption is specific to one type of neuron, the complex cells, which, as we reveal, normally match after the simple cells. Early environmental enrichment completely rescues the deficit by inducing histone acetylation and consequently advancing the matching process to coincide with the precocious plasticity. Our experiments thus demonstrate that the proper timing of the critical period is essential for establishing normal binocularity and the detrimental impact of its genetic misregulation can be ameliorated by environmental manipulations via epigenetic mechanisms.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures

References
-
- Alonso JM, Martinez LM. Functional connectivity between simple cells and complex cells in cat striate cortex. Nat Neurosci. 1998;1:395–403. - PubMed
-
- Baroncelli L, Sale A, Viegi A, Maya Vetencourt JF, De Pasquale R, Baldini S, Maffei L. Experience-dependent reactivation of ocular dominance plasticity in the adult visual cortex. Exp Neurol. 2010;226:100–109. - PubMed
-
- Begenisic T, Spolidoro M, Braschi C, Baroncelli L, Milanese M, Pietra G, Fabbri ME, Bonanno G, Cioni G, Maffei L, Sale A. Environmental enrichment decreases GABAergic inhibition and improves cognitive abilities, synaptic plasticity, and visual functions in a mouse model of Down syndrome. Front Cell Neurosci. 2011;5:29. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

