Endogenous oxytocin is necessary for preferential Fos expression to male odors in the bed nucleus of the stria terminalis in female Syrian hamsters
- PMID: 24012945
- PMCID: PMC3857696
- DOI: 10.1016/j.yhbeh.2013.08.016
Endogenous oxytocin is necessary for preferential Fos expression to male odors in the bed nucleus of the stria terminalis in female Syrian hamsters
Abstract
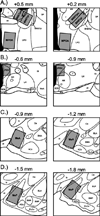
Successful reproduction in mammals depends on proceptive or solicitational behaviors that enhance the probability of encountering potential mates. In female Syrian hamsters, one such behavior is vaginal scent marking. Recent evidence suggests that the neuropeptide oxytocin (OT) may be critical for regulating this behavior. Blockade of OT receptors in the bed nucleus of the stria terminalis (BNST) or the medial preoptic area (MPOA) decreases vaginal marking responses to male odors; lesion data suggest that BNST, rather than MPOA, mediates this effect. However, how OT interacts with sexual odor processing to drive preferential solicitation is not known. To address this issue, intact female Syrian hamsters were exposed to male or female odors and their brains processed for immunohistochemistry for Fos, a marker of recent neuronal activation, and OT. Additional females were injected intracerebroventricularly (ICV) with an oxytocin receptor antagonist (OTA) or vehicle, and then tested for vaginal marking and Fos responses to sexual odors. Colocalization of OT and Fos in the paraventricular nucleus of the hypothalamus was unchanged following exposure to male odors, but decreased following exposure to female odors. Following injections of OTA, Fos expression to male odors was decreased in BNST, but not in MPOA or the medial amygdala (MA). Fos expression in BNST may be functionally relevant for vaginal marking, given that there was a positive correlation between Fos expression and vaginal marking for BNST, but not MPOA or MA. Together, these data suggest that OT facilitation of neuronal activity in BNST underlies the facilitative effects of OT on solicitational responses to male odors.
Keywords: Appetitive; Chemosensory; Olfaction; Precopulatory; Sexual motivation.
© 2013.
Figures


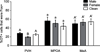
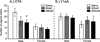


References
-
- Arakawa H, Arakawa K, Deak T. Oxytocin and vasopressin in the medial amygdala differentially modulate approach and avoidance behavior toward illness-related social odor. Neuroscience. 2010;171:1141–1151. - PubMed
-
- Beach FA. Sexual attractivity, proceptivity, and receptivity in female mammals. Horm. Behav. 1976;7:105–138. - PubMed
-
- Bennett AL, Greco B, Blasberg ME, Blaustein JD. Response to male odours in progestin receptor- and oestrogen receptor-containing cells in female rat brain. J. Neuroendocrinol. 2002;14:442–449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

