Indirect estimation of the comparative treatment effect in pharmacogenomic subgroups
- PMID: 24015225
- PMCID: PMC3754999
- DOI: 10.1371/journal.pone.0072256
Indirect estimation of the comparative treatment effect in pharmacogenomic subgroups
Abstract
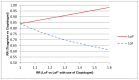
Evidence of clinical utility is a key issue in translating pharmacogenomics into clinical practice. Appropriately designed randomized controlled trials generally provide the most robust evidence of the clinical utility, but often only data from a pharmacogenomic association study are available. This paper details a method for reframing the results of pharmacogenomic association studies in terms of the comparative treatment effect for a pharmacogenomic subgroup to provide greater insight into the likely clinical utility of a pharmacogenomic marker, its' likely cost effectiveness, and the value of undertaking the further (often expensive) research required for translation into clinical practice. The method is based on the law of total probability, which relates marginal and conditional probability. It takes as inputs: the prevalence of the pharmacogenomic marker in the patient group of interest, prognostic effect of the pharmacogenomic marker based on observational association studies, and the unstratified comparative treatment effect based on one or more conventional randomized controlled trials. The critical assumption is that of exchangeability across the included studies. The method is demonstrated using a case study of cytochrome P450 (CYP) 2C19 genotype and the anti-platelet agent clopidogrel. Indirect subgroup analysis provided insight into relationship between the clinical utility of genotyping CYP2C19 and the risk ratio of cardiovascular outcomes between CYP2C19 genotypes for individuals using clopidogrel. In this case study the indirect and direct estimates of the treatment effect for the cytochrome P450 2C19 subgroups were similar. In general, however, indirect estimates are likely to have substantially greater risk of bias than an equivalent direct estimate.
Conflict of interest statement
Figures

References
-
- Lesko LJ, Zineh I, Huang SM (2010) What is clinical utility and why should we care? Clin Pharmacol Ther 88: 729–733. - PubMed
-
- Bucher HC, Guyatt GH, Griffith LE, Walter SD (1997) The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol 50: 683–691. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

