Very small embryonic-like stem cells (VSELs) represent a real challenge in stem cell biology: recent pros and cons in the midst of a lively debate
- PMID: 24018851
- PMCID: PMC3948156
- DOI: 10.1038/leu.2013.255
Very small embryonic-like stem cells (VSELs) represent a real challenge in stem cell biology: recent pros and cons in the midst of a lively debate
Abstract
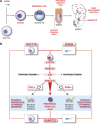
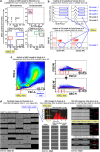
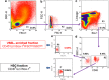
The concept that adult tissue, including bone marrow (BM), contains early-development cells with broader differentiation potential has again been recently challenged. In response, we would like to review the accumulated evidence from several independent laboratories that adult tissues, including BM, harbor a population of very rare stem cells that may cross germ layers in their differentiation potential. Thus, the BM stem cell compartment hierarchy needs to be revisited. These dormant, early-development cells that our group described as very small embryonic-like stem cells (VSELs) most likely overlap with similar populations of stem cells that have been identified in adult tissues by other investigators as the result of various experimental strategies and have been given various names. As reported, murine VSELs have some pluripotent stem cell characteristics. Moreover, they display several epiblast/germline markers that suggest their embryonic origin and developmental deposition in adult BM. Moreover, at the molecular level, changes in expression of parentally imprinted genes (for example, Igf2-H19) and resistance to insulin/insulin-like growth factor signaling (IIS) regulates their quiescent state in adult tissues. In several emergency situations related to organ damage, VSELs can be activated and mobilized into peripheral blood, and in appropriate animal models they contribute to tissue organ/regeneration. Interestingly, their number correlates with lifespan in mice, and they may also be involved in some malignancies. VSELs have been successfully isolated in several laboratories; however, some investigators experience problems with their isolation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Very small embryonic/epiblast-like stem cells (VSELs) and their potential role in aging and organ rejuvenation--an update and comparison to other primitive small stem cells isolated from adult tissues.Aging (Albany NY). 2012 Apr;4(4):235-46. doi: 10.18632/aging.100449. Aging (Albany NY). 2012. PMID: 22498452 Free PMC article. Review.
-
Molecular characterization of isolated from murine adult tissues very small embryonic/epiblast like stem cells (VSELs).Mol Cells. 2010 Jun;29(6):533-8. doi: 10.1007/s10059-010-0081-4. Epub 2010 Jun 4. Mol Cells. 2010. PMID: 20526817 Review.
-
Very small embryonic-like stem cells: characterization, developmental origin, and biological significance.Exp Hematol. 2008 Jun;36(6):742-51. doi: 10.1016/j.exphem.2008.03.010. Exp Hematol. 2008. PMID: 18474305 Free PMC article. Review.
-
scRNA-seq revealed transcriptional signatures of human umbilical cord primitive stem cells and their germ lineage origin regulated by imprinted genes.Sci Rep. 2024 Nov 26;14(1):29264. doi: 10.1038/s41598-024-79810-4. Sci Rep. 2024. PMID: 39587190 Free PMC article.
-
Molecular signature of adult bone marrow-purified very small embryonic-like stem cells supports their developmental epiblast/germ line origin.Leukemia. 2010 Aug;24(8):1450-61. doi: 10.1038/leu.2010.121. Epub 2010 May 27. Leukemia. 2010. PMID: 20508611
Cited by
-
Stem cells, progenitors & regenerative medicine: A retrospection.Indian J Med Res. 2015 Feb;141(2):154-61. doi: 10.4103/0971-5916.155543. Indian J Med Res. 2015. PMID: 25900949 Free PMC article. No abstract available.
-
Characterization and Isolation of Very Small Embryonic-like (VSEL) Stem Cells Obtained from Various Human Hematopoietic Cell Sources.Stem Cell Rev Rep. 2019 Oct;15(5):730-742. doi: 10.1007/s12015-019-09896-1. Stem Cell Rev Rep. 2019. PMID: 31172457
-
Very small embryonic-like stem-cell optimization of isolation protocols: an update of molecular signatures and a review of current in vivo applications.Exp Mol Med. 2013 Nov 15;45(11):e56. doi: 10.1038/emm.2013.117. Exp Mol Med. 2013. PMID: 24232255 Free PMC article. Review.
-
Proteomic Analysis of Murine Bone Marrow Very Small Embryonic-like Stem Cells at Steady-State Conditions and after In Vivo Stimulation by Nicotinamide and Follicle-Stimulating Factor Reflects their Germ-Lineage Origin and Multi Germ Layer Differentiation Potential.Stem Cell Rev Rep. 2023 Jan;19(1):120-132. doi: 10.1007/s12015-022-10445-6. Epub 2022 Aug 20. Stem Cell Rev Rep. 2023. PMID: 35986128 Free PMC article.
-
Autologous activated platelet-rich plasma injection into adult human ovary tissue: molecular mechanism, analysis, and discussion of reproductive response.Biosci Rep. 2019 Jun 4;39(6):BSR20190805. doi: 10.1042/BSR20190805. Print 2019 Jun 28. Biosci Rep. 2019. PMID: 31092698 Free PMC article. Review.
References
-
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006; 126: 663–676. - PubMed
-
- Zhao T, Zhang ZN, Rong Z, Xu Y. Immunogenicity of induced pluripotent stem cells. Nature 2011; 474: 212–215. - PubMed
-
- Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 2008; 451: 141–146. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

