Calcium signalling of human pluripotent stem cell-derived cardiomyocytes
- PMID: 24018947
- PMCID: PMC3936367
- DOI: 10.1113/jphysiol.2013.256495
Calcium signalling of human pluripotent stem cell-derived cardiomyocytes
Abstract
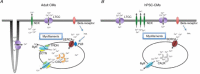
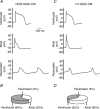
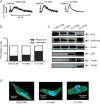
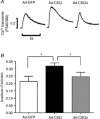
Loss of cardiomyocytes (CMs), which lack the innate ability to regenerate, due to ageing or pathophysiological conditions (e.g. myocardial infarction or MI) is generally considered irreversible, and can lead to conditions from cardiac arrhythmias to heart failure. Human (h) pluripotent stem cells (PSCs), including embryonic stem cells (ESC) and induced pluripotent stem cells (iPSCs), can self-renew while maintaining their pluripotency to differentiate into all cell types, including CMs. Therefore, hPSCs provide a potential unlimited ex vivo source of human CMs for disease modelling, drug discovery, cardiotoxicity screening and cell-based heart therapies. As a fundamental property of working CMs, Ca(2+) signalling and its role in excitation-contraction coupling are well described. However, the biology of these processes in hPSC-CMs is just becoming understood. Here we review what is known about the immature Ca(2+)-handling properties of hPSC-CMs, at the levels of global transients and sparks, and the underlying molecular basis in relation to the development of various in vitro approaches to drive their maturation.
Figures


References
-
- Anderson D, Self T, Mellor IR, Goh G, Hill SJ, Denning C. Transgenic enrichment of cardiomyocytes from human embryonic stem cells. Mol Ther. 2007;15:2027–2036. - PubMed
-
- Bassani JWM, Yuan WL, Bers DM. Fractional SR Ca release is regulated by trigger Ca and SR Ca content in cardiac myocytes. Am J Physiol Cell Physiol. 1995;268:C1313–C1319. - PubMed
-
- Beard NA, Laver DR, Dulhunty AF. Calsequestrin and the calcium release channel of skeletal and cardiac muscle. Prog Biophys Mol Biol. 2004;85:33–69. - PubMed
-
- Bers DM. Cardiac excitation–contraction coupling. Nature. 2002;415:198–205. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

