Adenoviral-mediated placental gene transfer of IGF-1 corrects placental insufficiency via enhanced placental glucose transport mechanisms
- PMID: 24019972
- PMCID: PMC3760855
- DOI: 10.1371/journal.pone.0074632
Adenoviral-mediated placental gene transfer of IGF-1 corrects placental insufficiency via enhanced placental glucose transport mechanisms
Abstract
Previous work in our laboratory demonstrated that over-expression of human insulin-like growth factor -1 (hIGF-1) in the placenta corrects fetal weight deficits in mouse, rat, and rabbit models of intrauterine growth restriction without changes in placental weight. The underlying mechanisms of this effect have not been elucidated. To investigate the effect of intra-placental IGF-1 over-expression on placental function we examined glucose transporter expression and localization in both a mouse model of IUGR and a model of human trophoblast, the BeWo Choriocarcinoma cell line.
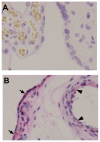
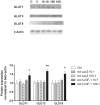
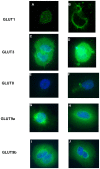
Methods: At gestational day 18, animals were divided into four groups; sham-operated controls, uterine artery branch ligation (UABL), UABL+Ad-hIGF-1 (10(8) PFU), UABL+Ad-LacZ (10(8) PFU). At gestational day 20, pups and placentas were harvested by C-section. For human studies, BeWo choriocarcinoma cells were grown in F12 complete medium +10%FBS. Cells were incubated in serum-free control media ± Ad-IGF-1 or Ad-LacZ for 48 hours. MOIs of 10∶1 and 100∶1 were utilized. The RNA, protein expression and localization of glucose transporters GLUT1, 3, 8, and 9 were analyzed by RT-PCR, Western blot and immunohistochemistry.
Results: In both the mouse placenta and BeWo, GLUT1 regulation was linked to altered protein localization. GLUT3, localized to the mouse fetal endothelial cells, was reduced in placental insufficiency but maintained with Ad-I GF-1 treatment. Interestingly, GLUT8 expression was reduced in the UABL placenta but up-regulated following Ad-IGF-1 in both mouse and human systems. GLUT9 expression in the mouse was increased by Ad-IGF-1 but this was not reflected in the BeWo, where Ad-IGF-1 caused moderate membrane relocalization.
Conclusion: Enhanced GLUT isoform transporter expression and relocalization to the membrane may be an important mechanism in Ad-hIGF-1mediated correction of placental insufficiency.
Conflict of interest statement
Figures



References
-
- Joost HG, Bell GI, Best JD, Birnbaum MJ, Charron MJ, et al. (2002) Nomenclature of the GLUT/SLC2A family of sugar/polyol transport facilitators. Am J Physiol Endocrinol Metab. 282: E974–976. - PubMed
-
- Illsley NP (2000) Glucose transporters in the human placenta. Placenta 21: 14–22. - PubMed
-
- Limesand SW, Regnault TR, Hay WW Jr (2004) Characterization of glucose transporter 8 (GLUT8) in the ovine placenta of normal and growth restricted fetuses. Placenta 25: 70–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

