A critical test of the "tunneling and coupled motion" concept in enzymatic alcohol oxidation
- PMID: 24020836
- PMCID: PMC3818283
- DOI: 10.1021/ja405917m
A critical test of the "tunneling and coupled motion" concept in enzymatic alcohol oxidation
Abstract
The physical mechanism of C-H bond activation by enzymes is the subject of intense study, and we have tested the predictions of two competing models for C-H activation in the context of alcohol dehydrogenase. The kinetic isotope effects (KIEs) in this enzyme have previously suggested a model of quantum mechanical tunneling and coupled motion of primary (1°) and secondary (2°) hydrogens. Here we measure the 2° H/T KIEs with both H and D at the 1° position and find that the 2° KIE is significantly deflated with D-transfer, consistent with the predictions of recent Marcus-like models of H-transfer. The results suggest that the fast dynamics of H-tunneling result in a 1° isotope effect on the structure of the tunneling ready state: the trajectory of D-transfer goes through a shorter donor-acceptor distance than that of H-transfer.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

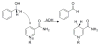
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

