Neuroprotective effects of perflurocarbon (oxycyte) after contusive spinal cord injury
- PMID: 24025081
- PMCID: PMC3904515
- DOI: 10.1089/neu.2013.3037
Neuroprotective effects of perflurocarbon (oxycyte) after contusive spinal cord injury
Abstract
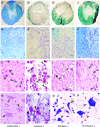
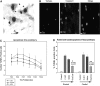
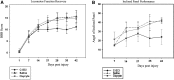
Spinal cord injury (SCI) often results in irreversible and permanent neurological deficits and long-term disability. Vasospasm, hemorrhage, and loss of microvessels create an ischemic environment at the site of contusive or compressive SCI and initiate the secondary injury cascades leading to progressive tissue damage and severely decreased functional outcome. Although the initial mechanical destructive events cannot be reversed, secondary injury damage occurs over several hours to weeks, a time frame during which therapeutic intervention could be achieved. One essential component of secondary injury cascade is the reduction in spinal cord blood flow with resultant decrease in oxygen delivery. Our group has recently shown that administration of fluorocarbon (Oxycyte) significantly increased parenchymal tissue oxygen levels during the usual postinjury hypoxic phase, and fluorocarbon has been shown to be effective in stroke and head injury. In the current study, we assessed the beneficial effects of Oxycyte after a moderate-to-severe contusion SCI was simulated in adult Long-Evans hooded rats. Histopathology and immunohistochemical analysis showed that the administration of 5 mL/kg of Oxycyte perfluorocarbon (60% emulsion) after SCI dramatically reduced destruction of spinal cord anatomy and resulted in a marked decrease of lesion area, less cell death, and greater white matter sparing at 7 and 42 days postinjury. Terminal deoxynucleotidyl transferase dUTP nick end labeling staining showed a significant reduced number of apoptotic cells in Oxycyte-treated animals, compared to the saline group. Collectively, these results demonstrate the potential neuroprotective effect of Oxycyte treatment after SCI, and its beneficial effects may be, in part, a result of reducing apoptotic cell death and tissue sparing. Further studies to determine the most efficacious Oxycyte dose and its mechanisms of protection are warranted.
Figures


References
-
- Bunge R.P., Puckett W.R., Becerra J.L., Marcillo A., and Quencer R.M. (1993). Observations on the pathology of human spinal cord injury. A review and classification of 22 new cases with details from a case of chronic cord compression with extensive focal demyelination. Adv. Neurol. 59, 75–89 - PubMed
-
- Baptiste D.C., and Fehlings M.G. (2007). Update on the treatment of spinal cord injury. Prog. Brain Res. 161, 217–233 - PubMed
-
- Tator C.H. (2006). Review of treatment trials in human spinal cord injury: issues, difficulties, and recommendations. J. Neurosurg. 59, 957–987 - PubMed
-
- Kwon B.K., Tetzlaff W., Grauer J.N., Beiner J., and Vaccaro A.R. (2004). Pathophysiology and pharmacologic treatment of acute spinal cord injury. Spine J. 4, 451–464 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

