Recreating kidney progenitors from pluripotent cells
- PMID: 24026757
- PMCID: PMC6219987
- DOI: 10.1007/s00467-013-2592-7
Recreating kidney progenitors from pluripotent cells
Abstract
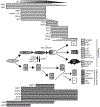
Access to human pluripotent cells theoretically provides a renewable source of cells that can give rise to any required cell type for use in cellular therapy or bioengineering. However, successfully directing this differentiation remains challenging for most desired endpoints cell type, including renal cells. This challenge is compounded by the difficulty in identifying the required cell type in vitro and the multitude of renal cell types required to build a kidney. Here we review our understanding of how the embryo goes about specifying the cells of the kidney and the progress to date in adapting this knowledge for the recreation of nephron progenitors and their mature derivatives from pluripotent cells.
Figures
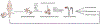
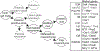
Similar articles
-
A strategy for generating kidney organoids: Recapitulating the development in human pluripotent stem cells.Dev Biol. 2016 Dec 15;420(2):210-220. doi: 10.1016/j.ydbio.2016.08.024. Epub 2016 Aug 23. Dev Biol. 2016. PMID: 27565022 Free PMC article. Review.
-
Recapitulating kidney development: Progress and challenges.Semin Cell Dev Biol. 2019 Jul;91:153-168. doi: 10.1016/j.semcdb.2018.08.015. Epub 2018 Sep 20. Semin Cell Dev Biol. 2019. PMID: 30184476 Free PMC article. Review.
-
Generation of Functioning Nephrons by Implanting Human Pluripotent Stem Cell-Derived Kidney Progenitors.Stem Cell Reports. 2018 Mar 13;10(3):766-779. doi: 10.1016/j.stemcr.2018.01.008. Epub 2018 Feb 8. Stem Cell Reports. 2018. PMID: 29429961 Free PMC article.
-
A Modular Differentiation System Maps Multiple Human Kidney Lineages from Pluripotent Stem Cells.Cell Rep. 2020 Apr 7;31(1):107476. doi: 10.1016/j.celrep.2020.03.040. Cell Rep. 2020. PMID: 32268094
-
Pluripotent Stem Cells to Rebuild a Kidney: The Lymph Node as a Possible Developmental Niche.Cell Transplant. 2016;25(6):1007-23. doi: 10.3727/096368915X688632. Epub 2015 Jul 8. Cell Transplant. 2016. PMID: 26160801 Review.
Cited by
-
Vascular deficiencies in renal organoids and ex vivo kidney organogenesis.Dev Biol. 2021 Sep;477:98-116. doi: 10.1016/j.ydbio.2021.04.009. Epub 2021 May 15. Dev Biol. 2021. PMID: 34000274 Free PMC article.
-
Directing human embryonic stem cell differentiation towards a renal lineage generates a self-organizing kidney.Nat Cell Biol. 2014 Jan;16(1):118-26. doi: 10.1038/ncb2894. Epub 2013 Dec 15. Nat Cell Biol. 2014. PMID: 24335651
-
Generating kidney tissue from pluripotent stem cells.Cell Death Discov. 2016 Jul 18;2:16053. doi: 10.1038/cddiscovery.2016.53. eCollection 2016. Cell Death Discov. 2016. PMID: 27551541 Free PMC article. Review.
-
Bioengineering and Stem Cell Technology in the Treatment of Congenital Heart Disease.J Clin Med. 2015 Apr 22;4(4):768-81. doi: 10.3390/jcm4040768. J Clin Med. 2015. PMID: 26239354 Free PMC article. Review.
-
Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips.Sci Rep. 2016 Oct 11;6:34845. doi: 10.1038/srep34845. Sci Rep. 2016. PMID: 27725720 Free PMC article.
References
-
- Bertram JF, Douglas-Denton RN, Diouf B, Hughson MD, Hoy WE. (2011) Human nephron number: implications for health and disease. Pediatr Nephrol 26(9):1529–33. - PubMed
-
- Mironov V, Drake C, Wen X. (2006) Research project: Charleston Bioengineered Kidney Project. Biotechnol J 1(9):903–905. - PubMed
-
- Skardal A, Zhang JX, Prestwich GD (2010) Bioprinting vessel-like constructs using hyaluronan hydrogels crosslinked with tetrahedral polyethylene glycol tetracrylates. Biomaterials 31: 6173–6181. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

