Screening drug-induced arrhythmia [corrected] using human induced pluripotent stem cell-derived cardiomyocytes and low-impedance microelectrode arrays
- PMID: 24030418
- PMCID: PMC3855862
- DOI: 10.1161/CIRCULATIONAHA.112.000570
Screening drug-induced arrhythmia [corrected] using human induced pluripotent stem cell-derived cardiomyocytes and low-impedance microelectrode arrays
Erratum in
- Circulation. 2014 Apr 15;129(15):e452
Abstract
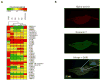
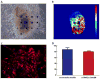
Background: Drug-induced arrhythmia is one of the most common causes of drug development failure and withdrawal from market. This study tested whether human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) combined with a low-impedance microelectrode array (MEA) system could improve on industry-standard preclinical cardiotoxicity screening methods, identify the effects of well-characterized drugs, and elucidate underlying risk factors for drug-induced arrhythmia. hiPSC-CMs may be advantageous over immortalized cell lines because they possess similar functional characteristics as primary human cardiomyocytes and can be generated in unlimited quantities.
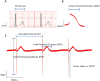
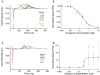
Methods and results: Pharmacological responses of beating embryoid bodies exposed to a comprehensive panel of drugs at 65 to 95 days postinduction were determined. Responses of hiPSC-CMs to drugs were qualitatively and quantitatively consistent with the reported drug effects in literature. Torsadogenic hERG blockers, such as sotalol and quinidine, produced statistically and physiologically significant effects, consistent with patch-clamp studies, on human embryonic stem cell-derived cardiomyocytes hESC-CMs. False-negative and false-positive hERG blockers were identified accurately. Consistent with published studies using animal models, early afterdepolarizations and ectopic beats were observed in 33% and 40% of embryoid bodies treated with sotalol and quinidine, respectively, compared with negligible early afterdepolarizations and ectopic beats in untreated controls.
Conclusions: We found that drug-induced arrhythmias can be recapitulated in hiPSC-CMs and documented with low impedance MEA. Our data indicate that the MEA/hiPSC-CM assay is a sensitive, robust, and efficient platform for testing drug effectiveness and for arrhythmia screening. This system may hold great potential for reducing drug development costs and may provide significant advantages over current industry standard assays that use immortalized cell lines or animal models.
Keywords: arrhythmias, cardiac; genomics; myocytes, cardiac; pharmacogenetics; pharmacology; stem cells.
Conflict of interest statement
Figures




References
-
- Braam SR, Tertoolen L, van de Stolpe A, Meyer T, Passier R, Mummery CL. Prediction of drug-induced cardiotoxicity using human embryonic stem cell-derived cardiomyocytes. Stem Cell Res. 2010;4:107–116. - PubMed
-
- Mandenius CF, Steel D, Noor F, Meyer T, Heinzle E, Asp J, Arain S, Kraushaar U, Bremer S, Class R, Sartipy P. Cardiotoxicity testing using pluripotent stem cell-derived human cardiomyocytes and state-of-the-art bioanalytics: A review. J Appl Toxicol. 2011;31:191–205. - PubMed
-
- Kannankeril PJ, Roden DM. Drug-induced long qt and torsade de pointes: Recent advances. Curr Opin Cardiol. 2007;22:39–43. - PubMed
-
- Denning C, Anderson D. Cardiomyocytes from human embryonic stem cells as predictors of cardiotoxicity. Drug Discovery Today: Ther Strategies. 2008;5(4):223–232.
-
- Fermini B, Fossa AA. The impact of drug-induced qt interval prolongation on drug discovery and development. Nat Rev Drug Discov. 2003;2:439–447. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

