Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex
- PMID: 24030490
- PMCID: PMC3819204
- DOI: 10.1126/science.1241475
Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex
Abstract
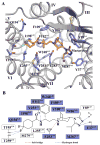
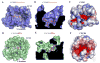
The CCR5 chemokine receptor acts as a co-receptor for HIV-1 viral entry. Here we report the 2.7 angstrom-resolution crystal structure of human CCR5 bound to the marketed HIV drug maraviroc. The structure reveals a ligand-binding site that is distinct from the proposed major recognition sites for chemokines and the viral glycoprotein gp120, providing insights into the mechanism of allosteric inhibition of chemokine signaling and viral entry. A comparison between CCR5 and CXCR4 crystal structures, along with models of co-receptor-gp120-V3 complexes, suggests that different charge distributions and steric hindrances caused by residue substitutions may be major determinants of HIV-1 co-receptor selectivity. These high-resolution insights into CCR5 can enable structure-based drug discovery for the treatment of HIV-1 infection.
Figures
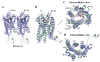
Comment in
-
Structural biology. A new bundle of prospects for blocking HIV-1 entry.Science. 2013 Sep 20;341(6152):1347-8. doi: 10.1126/science.1245384. Epub 2013 Sep 12. Science. 2013. PMID: 24030494 No abstract available.
References
-
- Baggiolini M. Chemokines and leukocyte traffic. Nature. 1998;392:565. - PubMed
-
- Gerard C, Rollins BJ. Chemokines and disease. Nat Immunol. 2001;2:108. - PubMed
-
- Blanpain C, et al. CCR5 binds multiple CC-chemokines: MCP-3 acts as a natural antagonist. Blood. 1999;94:1899. - PubMed
-
- Berger EA, Murphy PM, Farber JM. Chemokine receptors as HIV-1 coreceptors: roles in viral entry, tropism, and disease. Annu Rev Immunol. 1999;17:657. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

