Human brain stem structures respond differentially to noxious heat
- PMID: 24032012
- PMCID: PMC3764478
- DOI: 10.3389/fnhum.2013.00530
Human brain stem structures respond differentially to noxious heat
Abstract
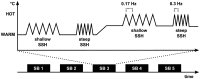
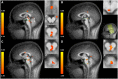
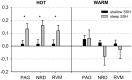
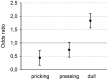
Concerning the physiological correlates of pain, the brain stem is considered to be one core region that is activated by noxious input. In animal studies, different slopes of skin heating (SSH) with noxious heat led to activation in different columns of the midbrain periaqueductal gray (PAG). The present study aimed at finding a method for differentiating structures in PAG and other brain stem structures, which are associated with different qualities of pain in humans according to the structures that were associated with different behavioral significances to noxious thermal stimulation in animals. Brain activity was studied by functional MRI in healthy subjects in response to steep and shallow SSH with noxious heat. We found differential activation to different SSH in the PAG and the rostral ventromedial medulla (RVM). In a second experiment, we demonstrate that the different SSH were associated with different pain qualities. Our experiments provide evidence that brainstem structures, i.e., the PAG and the RVM, become differentially activated by different SSH. Therefore, different SSH can be utilized when brain stem structures are investigated and when it is aimed to activate these structures differentially. Moreover, percepts of first pain were elicited by shallow SSH whereas percepts of second pain were elicited by steep SSH. The stronger activation of these brain stem structures to SSH, eliciting percepts of second vs. first pain, might be of relevance for activating different coping strategies in response to the noxious input with the two types of SSH.
Keywords: A-delta fiber; C-fiber; PAG; RVM; pain descriptors; periaqueductal grey; rostral ventromedial medulla; second pain.
Figures
Similar articles
-
Resolving the Brainstem Contributions to Attentional Analgesia.J Neurosci. 2017 Mar 1;37(9):2279-2291. doi: 10.1523/JNEUROSCI.2193-16.2016. Epub 2017 Jan 17. J Neurosci. 2017. PMID: 28096471 Free PMC article.
-
Offset analgesia is mediated by activation in the region of the periaqueductal grey and rostral ventromedial medulla.Neuroimage. 2009 Sep;47(3):1002-6. doi: 10.1016/j.neuroimage.2009.04.032. Epub 2009 Apr 16. Neuroimage. 2009. PMID: 19375510
-
Quantitative comparison of inhibition of visceral and cutaneous spinal nociceptive transmission from the midbrain and medulla in the rat.J Neurophysiol. 1987 Oct;58(4):850-65. doi: 10.1152/jn.1987.58.4.850. J Neurophysiol. 1987. PMID: 2824712
-
Descending antinociceptive mechanisms in the brainstem: their role in the animal's defensive system.J Physiol Paris. 1996;90(1):15-25. doi: 10.1016/0928-4257(96)87165-8. J Physiol Paris. 1996. PMID: 8803851 Review.
-
The activation of bulbo-spinal controls by peripheral nociceptive inputs: diffuse noxious inhibitory controls.Biol Res. 1995;28(1):113-25. Biol Res. 1995. PMID: 8728826 Review.
Cited by
-
Influence of acute pain on valence rating of words.PLoS One. 2021 Mar 18;16(3):e0248744. doi: 10.1371/journal.pone.0248744. eCollection 2021. PLoS One. 2021. PMID: 33735235 Free PMC article.
-
Investigating the human brainstem with structural and functional MRI.Front Hum Neurosci. 2014 Feb 28;8:116. doi: 10.3389/fnhum.2014.00116. eCollection 2014. Front Hum Neurosci. 2014. PMID: 24616692 Free PMC article. No abstract available.
-
Evidence- and data-driven classification of low back pain via artificial intelligence: Protocol of the PREDICT-LBP study.PLoS One. 2023 Aug 21;18(8):e0282346. doi: 10.1371/journal.pone.0282346. eCollection 2023. PLoS One. 2023. PMID: 37603539 Free PMC article.
-
Boundary effects of expectation in human pain perception.Sci Rep. 2019 Jul 1;9(1):9443. doi: 10.1038/s41598-019-45811-x. Sci Rep. 2019. PMID: 31263144 Free PMC article.
-
A 7-Tesla MRI study of the periaqueductal gray: resting state and task activation under threat.Soc Cogn Affect Neurosci. 2022 Feb 15;17(2):187-197. doi: 10.1093/scan/nsab085. Soc Cogn Affect Neurosci. 2022. PMID: 34244809 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

