Sweeten PAMPs: Role of Sugar Complexed PAMPs in Innate Immunity and Vaccine Biology
- PMID: 24032031
- PMCID: PMC3759294
- DOI: 10.3389/fimmu.2013.00248
Sweeten PAMPs: Role of Sugar Complexed PAMPs in Innate Immunity and Vaccine Biology
Abstract
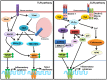
Innate sensors play a critical role in the early innate immune responses to invading pathogens through sensing of diverse biochemical signatures also known as pathogen associated molecular patterns (PAMPs). These biochemical signatures primarily consist of a major family of biomolecules such as proteins, lipids, nitrogen bases, and sugar and its complexes, which are distinct from host molecules and exclusively expressed in pathogens and essential to their survival. The family of sensors known as pattern recognition receptors (PRRs) are germ-line encoded, evolutionarily conserved molecules, and consist of Toll-like receptors (TLRs), RIG-I-like receptors (RLRs), NOD-like receptors (NLRs), C-type lectin-like receptors (CLRs), and DNA sensors. Sensing of PAMP by PRR initiates the cascade of signaling leading to the activation of transcription factors, such as NF-κB and interferon regulatory factors (IRFs), resulting in a variety of cellular responses, including the production of interferons (IFNs) and pro-inflammatory cytokines. In this review, we discuss sensing of different types of glycosylated PAMPs such as β-glucan (a polymeric sugar) or lipopolysaccharides, nucleic acid, and so on (sugar complex PAMPs) by different families of sensors, its role in pathogenesis, and its application in development of potential vaccine and vaccine adjuvants.
Keywords: disease pathogenesis; innate immunity; innate sensors; sugar associated PAMPs; vaccinology.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

