Assembly of bio-nanoparticles for double controlled drug release
- PMID: 24040316
- PMCID: PMC3765395
- DOI: 10.1371/journal.pone.0074679
Assembly of bio-nanoparticles for double controlled drug release
Abstract
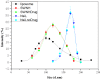
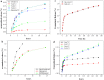
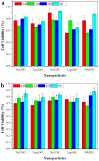
A critical limiting factor of chemotherapy is the unacceptably high toxicity. The use of nanoparticle based drug carriers has significantly reduced the side effects and facilitated the delivery of drugs. Source of the remaining side effect includes (1) the broad final in vivo distribution of the administrated nanoparticles, and (2) strong basal drug release from nanoparticles before they could reach the tumor. Despite the advances in pH-triggered release, undesirable basal drug release has been a constant challenge under in vivo conditions. In this study, functionalized single walled carbon nanohorn supported immunoliposomes were assembled for paclitaxel delivery. The immunoliposomes were formulated with polyethylene glycol, thermal stable and pH sensitive phospholipids. Each nanohorn was found to be encapsulated within one immunoliposome. Results showed a highly pH dependent release of paclitaxel in the presence of serum at body temperature with minimal basal release under physiological conditions. Upon acidification, paclitaxel was released at a steady rate over 30 days with a cumulative release of 90% of the loaded drug. The drug release results proved our hypothesized double controlled release mechanism from the nanoparticles. Other results showed the nanoparticles have doubled loading capacity compared to that of traditional liposomes and higher affinity to breast cancer cells overexpressing Her2 receptors. Internalized nanoparticles were found in lysosomes.
Conflict of interest statement
Figures




References
-
- Brigger I, Dubernet C, Couvreur P (2002) Nanoparticles in cancer therapy and diagnosis. Adv Drug Deliv Rev 54: 631–651. - PubMed
-
- Parveen S, Mistra R, Sahoo SK (2012) Nanoparticles: a boon to drug delivery, therapeutics, diagnostics and imaging. Nanomedicine 8: 147–166. - PubMed
-
- Reddy LH (2005) Drug delivery to tumors: recent strategies. J Pharm Pharmacol 37: 1231–1242. - PubMed
-
- Yang T, Choi M, Cui F, Kim JS, Chung S, et al. (2007) Preparation and evaluation of paclitaxel-loaded PEGylated immunoliposome. J Control Release 120: 169–177. - PubMed
-
- Ptail Y, Sadhukha T, Ma L, Panyam J (2009) Nanoparticle-mediated simultaneous and targeted delivery of paclitaxel and tarquidar overcomes tumor drug resistance. J Control Release 136: 21–29. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

