Autocrine TNF-α production supports CML stem and progenitor cell survival and enhances their proliferation
- PMID: 24041577
- PMCID: PMC3953090
- DOI: 10.1182/blood-2013-02-485607
Autocrine TNF-α production supports CML stem and progenitor cell survival and enhances their proliferation
Abstract
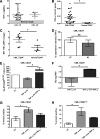
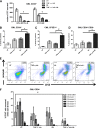
Chronic myeloid leukemia (CML) stem cells are not dependent on BCR-ABL kinase for their survival, suggesting that kinase-independent mechanisms must contribute to their persistence. We observed that CML stem/progenitor cells (SPCs) produce tumor necrosis factor-α (TNF-α) in a kinase-independent fashion and at higher levels relative to their normal counterparts. We therefore investigated the role of TNF-α and found that it supports survival of CML SPCs by promoting nuclear factor κB/p65 pathway activity and expression of the interleukin 3 and granulocyte/macrophage-colony stimulating factor common β-chain receptor. Furthermore, we demonstrate that in CML SPCs, inhibition of autocrine TNF-α signaling via a small-molecule TNF-α inhibitor induces apoptosis. Moreover TNF-α inhibition combined with nilotinib induces significantly more apoptosis relative to either treatment alone and a reduction in the absolute number of primitive quiescent CML stem cells. These results highlight a novel survival mechanism of CML SPCs and suggest a new putative therapeutic target for their eradication.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

