Soluble epoxide hydrolase inhibitor trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid is neuroprotective in rat model of ischemic stroke
- PMID: 24043255
- PMCID: PMC3882464
- DOI: 10.1152/ajpheart.00471.2013
Soluble epoxide hydrolase inhibitor trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid is neuroprotective in rat model of ischemic stroke
Abstract
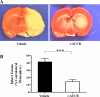
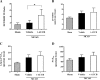
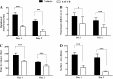
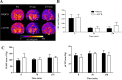
Soluble epoxide hydrolase (sEH) diminishes vasodilatory and neuroprotective effects of epoxyeicosatrienoic acids by hydrolyzing them to inactive dihydroxy metabolites. The primary goals of this study were to investigate the effects of acute sEH inhibition by trans-4-[4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid (t-AUCB) on infarct volume, functional outcome, and changes in cerebral blood flow (CBF) in a rat model of ischemic stroke. Focal cerebral ischemia was induced in rats for 90 min followed by reperfusion. At the end of 24 h after reperfusion rats were euthanized for infarct volume assessment by triphenyltetrazolium chloride staining. Brain cortical sEH activity was assessed by ultra performance liquid chromatography-tandem mass spectrometry. Functional outcome at 24 and 48 h after reperfusion was evaluated by arm flexion and sticky-tape tests. Changes in CBF were assessed by arterial spin-labeled-MRI at baseline, during ischemia, and at 180 min after reperfusion. Neuroprotective effects of t-AUCB were evaluated in primary rat neuronal cultures by Cytotox-Flour kit and propidium iodide staining. t-AUCB significantly reduced cortical infarct volume by 35% (14.5 ± 2.7% vs. 41.5 ± 4.5%), elevated cumulative epoxyeicosatrienoic acids-to-dihydroxyeicosatrienoic acids ratio in brain cortex by twofold (4.40 ± 1.89 vs. 1.97 ± 0.85), and improved functional outcome in arm-flexion test (day 1: 3.28 ± 0.5 s vs. 7.50 ± 0.9 s; day 2: 1.71 ± 0.4 s vs. 5.28 ± 0.5 s) when compared with that of the vehicle-treated group. t-AUCB significantly reduced neuronal cell death in a dose-dependent manner (vehicle: 70.9 ± 7.1% vs. t-AUCB0.1μM: 58 ± 5.11% vs. t-AUCB0.5μM: 39.9 ± 5.8%). These findings suggest that t-AUCB may exert its neuroprotective effects by affecting multiple components of neurovascular unit including neurons, astrocytes, and microvascular flow.
Keywords: arachidonic acid; cerebral blood flow; cytochrome P-450; ischemic stroke; middle cerebral artery occlusion.
Figures


Similar articles
-
Inhibition of soluble epoxide hydrolase by trans-4- [4-(3-adamantan-1-yl-ureido)-cyclohexyloxy]-benzoic acid is protective against ischemia-reperfusion injury.J Cardiovasc Pharmacol. 2010 Jan;55(1):67-73. doi: 10.1097/FJC.0b013e3181c37d69. J Cardiovasc Pharmacol. 2010. PMID: 19834332 Free PMC article.
-
Role of haem oxygenase in the renoprotective effects of soluble epoxide hydrolase inhibition in diabetic spontaneously hypertensive rats.Clin Sci (Lond). 2013 Oct;125(7):349-59. doi: 10.1042/CS20130003. Clin Sci (Lond). 2013. PMID: 23611540
-
Soluble epoxide hydrolase inhibition improves coronary endothelial function and prevents the development of cardiac alterations in obese insulin-resistant mice.Am J Physiol Heart Circ Physiol. 2015 May 1;308(9):H1020-9. doi: 10.1152/ajpheart.00465.2014. Epub 2015 Feb 27. Am J Physiol Heart Circ Physiol. 2015. PMID: 25724490 Free PMC article.
-
Meloxicam fails to augment the reno-protective effects of soluble epoxide hydrolase inhibition in streptozotocin-induced diabetic rats via increased 20-HETE levels.Prostaglandins Other Lipid Mediat. 2017 Sep;132:3-11. doi: 10.1016/j.prostaglandins.2016.08.004. Epub 2016 Sep 3. Prostaglandins Other Lipid Mediat. 2017. PMID: 27596333 Review.
-
Soluble epoxide hydrolase: a new target for cardioprotection.Curr Opin Investig Drugs. 2009 Mar;10(3):253-8. Curr Opin Investig Drugs. 2009. PMID: 19333883 Free PMC article. Review.
Cited by
-
Stress, Genes, and Hypertension. Contribution of the ISIAH Rat Strain Study.Curr Hypertens Rep. 2018 Jun 16;20(8):66. doi: 10.1007/s11906-018-0870-2. Curr Hypertens Rep. 2018. PMID: 29909475 Review.
-
Soluble Epoxide Hydrolase Inhibitor Ameliorates Olfactory Dysfunction, Modulates Microglia Polarization, and Attenuates Neuroinflammation after Ischemic Brain Injury.J Neuroimmune Pharmacol. 2024 Oct 17;19(1):54. doi: 10.1007/s11481-024-10155-5. J Neuroimmune Pharmacol. 2024. PMID: 39417923
-
Probing changes in brain esterified oxylipin concentrations during the early stages of pathogenesis in Alzheimer's Disease transgenic rats.Neurosci Lett. 2022 Nov 20;791:136921. doi: 10.1016/j.neulet.2022.136921. Epub 2022 Oct 18. Neurosci Lett. 2022. PMID: 36270451 Free PMC article.
-
20-Hydroxyeicosatetraenoic Acid Inhibition by HET0016 Offers Neuroprotection, Decreases Edema, and Increases Cortical Cerebral Blood Flow in a Pediatric Asphyxial Cardiac Arrest Model in Rats.J Cereb Blood Flow Metab. 2015 Nov;35(11):1757-63. doi: 10.1038/jcbfm.2015.117. Epub 2015 Jun 10. J Cereb Blood Flow Metab. 2015. PMID: 26058691 Free PMC article.
-
Humble beginnings with big goals: Small molecule soluble epoxide hydrolase inhibitors for treating CNS disorders.Prog Neurobiol. 2019 Jan;172:23-39. doi: 10.1016/j.pneurobio.2018.11.001. Epub 2018 Nov 14. Prog Neurobiol. 2019. PMID: 30447256 Free PMC article. Review.
References
-
- Abe K, Kogure K, Yamamoto H, Imazawa M, Miyamoto K. Mechanism of arachidonic acid liberation during ischemia in gerbil cerebral cortex. J Neurochem 48: 503–509, 1987 - PubMed
-
- Alkayed NJ, Birks EK, Hudetz AG, Roman RJ, Henderson L, Harder DR. Inhibition of brain P-450 arachidonic acid epoxygenase decreases baseline cerebral blood flow. Am J Physiol Heart Circ Physiol 271: H1541–H1546, 1996 - PubMed
-
- Aronowski J, Samways E, Strong R, Rhoades HM, Grotta JC. An alternative method for the quantitation of neuronal damage after experimental middle cerebral artery occlusion in rats: analysis of behavioral deficit. J Cereb Blood Flow Metab 16: 705–713, 1996 - PubMed
-
- Baskaya MK, Prasad MR, Donaldson D, Hu Y, Rao AM, Dempsey RJ. Enhanced accumulation of free fatty acids in experimental focal cerebral ischemia. Prostaglandins Leukot Essent Fatty Acids 54: 167–171, 1996 - PubMed
-
- Bazan NG. Free arachidonic acid and other lipids in the nervous system during early ischemia and after electroshock. Adv Exp Med Biol 72: 317–335, 1976 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

