Interrogating biology with force: single molecule high-resolution measurements with optical tweezers
- PMID: 24047980
- PMCID: PMC3785866
- DOI: 10.1016/j.bpj.2013.08.007
Interrogating biology with force: single molecule high-resolution measurements with optical tweezers
Abstract
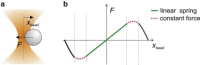
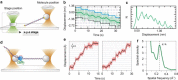
Single molecule force spectroscopy methods, such as optical and magnetic tweezers and atomic force microscopy, have opened up the possibility to study biological processes regulated by force, dynamics of structural conformations of proteins and nucleic acids, and load-dependent kinetics of molecular interactions. Among the various tools available today, optical tweezers have recently seen great progress in terms of spatial resolution, which now allows the measurement of atomic-scale conformational changes, and temporal resolution, which has reached the limit of the microsecond-scale relaxation times of biological molecules bound to a force probe. Here, we review different strategies and experimental configurations recently developed to apply and measure force using optical tweezers. We present the latest progress that has pushed optical tweezers' spatial and temporal resolution down to today's values, discussing the experimental variables and constraints that are influencing measurement resolution and how these can be optimized depending on the biological molecule under study.
Copyright © 2013 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

