A novel extracellular role for tissue transglutaminase in matrix-bound VEGF-mediated angiogenesis
- PMID: 24052076
- PMCID: PMC3789176
- DOI: 10.1038/cddis.2013.318
A novel extracellular role for tissue transglutaminase in matrix-bound VEGF-mediated angiogenesis
Abstract
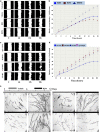
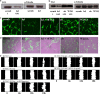
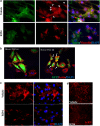
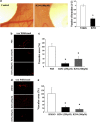
The importance of tissue transglutaminase (TG2) in angiogenesis is unclear and contradictory. Here we show that inhibition of extracellular TG2 protein crosslinking or downregulation of TG2 expression leads to inhibition of angiogenesis in cell culture, the aorta ring assay and in vivo models. In a human umbilical vein endothelial cell (HUVEC) co-culture model, inhibition of extracellular TG2 activity can halt the progression of angiogenesis, even when introduced after tubule formation has commenced and after addition of excess vascular endothelial growth factor (VEGF). In both cases, this leads to a significant reduction in tubule branching. Knockdown of TG2 by short hairpin (shRNA) results in inhibition of HUVEC migration and tubule formation, which can be restored by add back of wt TG2, but not by the transamidation-defective but GTP-binding mutant W241A. TG2 inhibition results in inhibition of fibronectin deposition in HUVEC monocultures with a parallel reduction in matrix-bound VEGFA, leading to a reduction in phosphorylated VEGF receptor 2 (VEGFR2) at Tyr¹²¹⁴ and its downstream effectors Akt and ERK1/2, and importantly its association with β1 integrin. We propose a mechanism for the involvement of matrix-bound VEGFA in angiogenesis that is dependent on extracellular TG2-related activity.
Figures



Similar articles
-
The functional relationship between transglutaminase 2 and transforming growth factor β1 in the regulation of angiogenesis and endothelial-mesenchymal transition.Cell Death Dis. 2017 Sep 7;8(9):e3032. doi: 10.1038/cddis.2017.399. Cell Death Dis. 2017. PMID: 28880274 Free PMC article.
-
The interaction of heparan sulfate proteoglycans with endothelial transglutaminase-2 limits VEGF165-induced angiogenesis.Sci Signal. 2015 Jul 14;8(385):ra70. doi: 10.1126/scisignal.aaa0963. Sci Signal. 2015. PMID: 26175493
-
Matrix changes induced by transglutaminase 2 lead to inhibition of angiogenesis and tumor growth.Cell Death Differ. 2006 Sep;13(9):1442-53. doi: 10.1038/sj.cdd.4401816. Epub 2005 Nov 18. Cell Death Differ. 2006. PMID: 16294209
-
Extracellular TG2: emerging functions and regulation.FEBS J. 2011 Dec;278(24):4704-16. doi: 10.1111/j.1742-4658.2011.08346.x. Epub 2011 Nov 21. FEBS J. 2011. PMID: 21902810 Free PMC article. Review.
-
Tissue transglutaminase: a multifunctional and multisite regulator in health and disease.Physiol Rev. 2024 Jan 1;104(1):281-325. doi: 10.1152/physrev.00003.2023. Epub 2023 Sep 15. Physiol Rev. 2024. PMID: 37712623 Review.
Cited by
-
Transglutaminase 2 has opposing roles in the regulation of cellular functions as well as cell growth and death.Cell Death Dis. 2016 Jun 2;7(6):e2244. doi: 10.1038/cddis.2016.150. Cell Death Dis. 2016. PMID: 27253408 Free PMC article. Review.
-
Corosolic acid: antiangiogenic activity and safety of intravitreal injection in rats eyes.Doc Ophthalmol. 2019 Jun;138(3):181-194. doi: 10.1007/s10633-019-09682-x. Epub 2019 Feb 26. Doc Ophthalmol. 2019. PMID: 30809742
-
Vasohibins: new transglutaminase-like cysteine proteases possessing a non-canonical Cys-His-Ser catalytic triad.Bioinformatics. 2016 May 15;32(10):1441-5. doi: 10.1093/bioinformatics/btv761. Epub 2016 Jan 21. Bioinformatics. 2016. PMID: 26794318 Free PMC article.
-
Transglutaminase 2: a novel therapeutic target for idiopathic pulmonary fibrosis using selective small molecule inhibitors.Amino Acids. 2021 Feb;53(2):205-217. doi: 10.1007/s00726-020-02938-w. Epub 2021 Jan 21. Amino Acids. 2021. PMID: 33474654 Free PMC article.
-
Endostatin and transglutaminase 2 are involved in fibrosis of the aging kidney.Kidney Int. 2016 Jun;89(6):1281-92. doi: 10.1016/j.kint.2016.01.030. Epub 2016 Apr 14. Kidney Int. 2016. PMID: 27165830 Free PMC article.
References
-
- Hellberg C, Ostman A, Heldin CH. PDGF and vessel maturation. Recent Results Cancer Res. 2010;180:103–114. - PubMed
-
- Siemerink MJ, Augustin AJ, Schlingemann RO. Mechanisms of ocular angiogenesis and its molecular mediators. Dev Ophthalmol. 2010;46:4–20. - PubMed
-
- Finn RS, Zhu AX. Targeting angiogenesis in hepatocellular carcinoma: focus on VEGF and bevacizumab. Expert Rev Anticancer Ther. 2009;9:503–509. - PubMed
-
- Roodhart JM, Langenberg MH, Witteveen E, Voest EE. The molecular basis of class side effects due to treatment with inhibitors of the VEGF/VEGFR pathway. Curr Clin Pharmacol. 2008;3:132–143. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

