Enzymatic activity of CaMKII is not required for its interaction with the glutamate receptor subunit GluN2B
- PMID: 24056996
- PMCID: PMC3834147
- DOI: 10.1124/mol.113.089045
Enzymatic activity of CaMKII is not required for its interaction with the glutamate receptor subunit GluN2B
Abstract
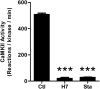
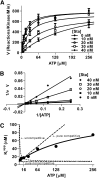
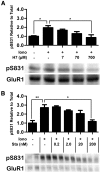
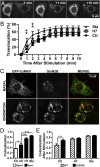
Binding of the Ca²⁺/calmodulin (CaM)-dependent protein kinase II (CaMKII) to the NMDA-type glutamate receptor subunit GluN2B is an important control mechanism for the regulation of synaptic strength. CaMKII binding to GluN2B and CaMKII translocation to synapses are induced by an initial Ca²⁺/CaM stimulus, which also activates the kinase. Indeed, several mechanistically different CaMKII inhibitors [tatCN21 and KN-93 (N-[2-[[[3-(4-chlorophenyl)-2-propenyl]methylamino]methyl]phenyl]-N-(2-hydroxyethyl)-4-methoxybenzenesulphonamide)] and inactivating mutations (K42M, A302R, and T305/T306D) impair this interaction, suggesting that it requires CaMKII enzymatic activity. However, this study shows that two general kinase inhibitors, H7 [1-(5-isoquinolinylsulfonyl)-2-methylpiperazine] and staurosporine (Sta), which inhibit CaMKII activity by yet another mechanism, did not interfere with GluN2B binding in vitro or within cells. In contrast to a previous report, we found that Sta, like H7, inhibited CaMKII in an ATP-competitive manner. Nucleotide binding significantly enhances CaMKII/GluN2B binding in vitro, but the nucleotide competition by H7 or Sta did not prevent this effect and instead even mimicked it. H7 (700 µM) and Sta (2 µM) efficiently blocked enzymatic activity of CaMKII, both in vitro and within cells. However, neither H7 nor Sta prevented Ca²⁺-induced translocation of CaMKII to GluN2B in heterologous cells or to synapses in hippocampal neurons. Thus, activity of CaMKII (or of any other kinase inhibited by H7 or Sta) is not required for stimulation-induced GluN2B-binding or synaptic translocation of CaMKII, despite previous indication to the contrary. This shows that results with inhibitors and inhibiting mutants can be caused by structural effects independent from catalytic activity, and that detailed understanding of the mechanisms is required for their interpretation.
Figures

References
-
- Barria A, Malinow R. (2005) NMDA receptor subunit composition controls synaptic plasticity by regulating binding to CaMKII. Neuron 48:289–301 - PubMed
-
- Bayer KU, De Koninck P, Leonard AS, Hell JW, Schulman H. (2001) Interaction with the NMDA receptor locks CaMKII in an active conformation. Nature 411:801–805 - PubMed
-
- Chen HX, Otmakhov N, Strack S, Colbran RJ, Lisman JE. (2001) Is persistent activity of calcium/calmodulin-dependent kinase required for the maintenance of LTP? J Neurophysiol 85:1368–1376 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

