Auxin and cytokinin control formation of the quiescent centre in the adventitious root apex of Arabidopsis
- PMID: 24061489
- PMCID: PMC3806543
- DOI: 10.1093/aob/mct215
Auxin and cytokinin control formation of the quiescent centre in the adventitious root apex of Arabidopsis
Abstract
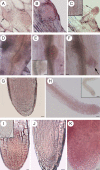
Background and aims: Adventitious roots (ARs) are part of the root system in numerous plants, and are required for successful micropropagation. In the Arabidopsis thaliana primary root (PR) and lateral roots (LRs), the quiescent centre (QC) in the stem cell niche of the meristem controls apical growth with the involvement of auxin and cytokinin. In arabidopsis, ARs emerge in planta from the hypocotyl pericycle, and from different tissues in in vitro cultured explants, e.g. from the stem endodermis in thin cell layer (TCL) explants. The aim of this study was to investigate the establishment and maintenance of the QC in arabidopsis ARs, in planta and in TCL explants, because information about this process is still lacking, and it has potential use for biotechnological applications.
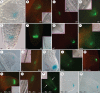
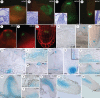
Methods: Expression of PR/LR QC markers and auxin influx (LAX3)/efflux (PIN1) genes was investigated in the presence/absence of exogenous auxin and cytokinin. Auxin was monitored by the DR5::GUS system and cytokinin by immunolocalization. The expression of the auxin-biosynthetic YUCCA6 gene was also investigated by in situ hybridization in planta and in AR-forming TCLs from the indole acetic acid (IAA)-overproducing superroot2-1 mutant and its wild type.
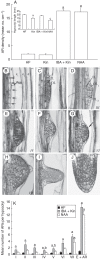
Key results: The accumulation of auxin and the expression of the QC marker WOX5 characterized the early derivatives of the AR founder cells, in planta and in in vitro cultured TCLs. By determination of PIN1 auxin efflux carrier and LAX3 auxin influx carrier activities, an auxin maximum was determined to occur at the AR tip, to which WOX5 expression was restricted, establishing the positioning of the QC. Cytokinin caused a restriction of LAX3 and PIN1 expression domains, and concomitantly the auxin biosynthesis YUCCA6 gene was expressed in the apex.
Conclusions: In ARs formed in planta and TCLs, the QC is established in a similar way, and auxin transport and biosynthesis are involved through cytokinin tuning.
Keywords: Adventitious root apex; Arabidopsis thaliana; WOX5; auxin biosynthesis; auxin transport; cytokinin localization; quiescent centre; root meristemoids; stem endodermis; thin cell layers.
Figures



Similar articles
-
Arabidopsis SHR and SCR transcription factors and AUX1 auxin influx carrier control the switch between adventitious rooting and xylogenesis in planta and in in vitro cultured thin cell layers.Ann Bot. 2015 Mar;115(4):617-28. doi: 10.1093/aob/mcu258. Epub 2015 Jan 23. Ann Bot. 2015. PMID: 25617411 Free PMC article.
-
The quiescent center and the stem cell niche in the adventitious roots of Arabidopsis thaliana.Plant Signal Behav. 2016 May 3;11(5):e1176660. doi: 10.1080/15592324.2016.1176660. Plant Signal Behav. 2016. PMID: 27089118 Free PMC article. Review.
-
Indole-3-butyric acid promotes adventitious rooting in Arabidopsis thaliana thin cell layers by conversion into indole-3-acetic acid and stimulation of anthranilate synthase activity.BMC Plant Biol. 2017 Jul 11;17(1):121. doi: 10.1186/s12870-017-1071-x. BMC Plant Biol. 2017. PMID: 28693423 Free PMC article.
-
Cytokinin induces cell division in the quiescent center of the Arabidopsis root apical meristem.Curr Biol. 2013 Oct 21;23(20):1979-89. doi: 10.1016/j.cub.2013.08.008. Epub 2013 Oct 10. Curr Biol. 2013. PMID: 24120642
-
Cytokinin-Controlled Gradient Distribution of Auxin in Arabidopsis Root Tip.Int J Mol Sci. 2021 Apr 8;22(8):3874. doi: 10.3390/ijms22083874. Int J Mol Sci. 2021. PMID: 33918090 Free PMC article. Review.
Cited by
-
Medicago truncatula root developmental changes by growth-promoting microbes isolated from Fabaceae, growing on organic farms, involve cell cycle changes and WOX5 gene expression.Planta. 2019 Nov 29;251(1):25. doi: 10.1007/s00425-019-03300-5. Planta. 2019. PMID: 31784832
-
Jasmonic Acid Methyl Ester Induces Xylogenesis and Modulates Auxin-Induced Xylary Cell Identity with NO Involvement.Int J Mol Sci. 2019 Sep 10;20(18):4469. doi: 10.3390/ijms20184469. Int J Mol Sci. 2019. PMID: 31510080 Free PMC article.
-
The auxin receptor TIR1 homolog (PagFBL 1) regulates adventitious rooting through interactions with Aux/IAA28 in Populus.Plant Biotechnol J. 2019 Feb;17(2):338-349. doi: 10.1111/pbi.12980. Epub 2018 Jul 24. Plant Biotechnol J. 2019. PMID: 29949229 Free PMC article.
-
A detailed expression map of the PIN1 auxin transporter in Arabidopsis thaliana root.BMC Plant Biol. 2016 Jan 27;16 Suppl 1(Suppl 1):5. doi: 10.1186/s12870-015-0685-0. BMC Plant Biol. 2016. PMID: 26821586 Free PMC article.
-
Enhancing auxin accumulation in maize root tips improves root growth and dwarfs plant height.Plant Biotechnol J. 2018 Jan;16(1):86-99. doi: 10.1111/pbi.12751. Epub 2017 Jun 23. Plant Biotechnol J. 2018. PMID: 28499064 Free PMC article.
References
-
- Aloni R, Langhans M, Aloni E, Dreieicher E, Ullrich CI. Root-synthesized cytokinin in Arabidopsis is distributed in the shoot by the transpiration stream. Journal of Experimental Botany. 2005;56:1535–1544. - PubMed
-
- Benková E, Michniewicz M, Sauer M, et al. Local, efflux-dependent auxin gradients as a common module for plant organ formation. Cell. 2003;115:591–602. - PubMed
-
- van den Berg C, Willemsen V, Hendriks G, Weisbeek P, Scheres B. Short-range control of cell differentiation in the Arabidopsis root meristem. Nature. 1997;390:287–289. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

