P53 regulates the migration of mesenchymal stromal cells in response to the tumor microenvironment through both CXCL12-dependent and -independent mechanisms
- PMID: 24064862
- PMCID: PMC3834256
- DOI: 10.3892/ijo.2013.2109
P53 regulates the migration of mesenchymal stromal cells in response to the tumor microenvironment through both CXCL12-dependent and -independent mechanisms
Abstract
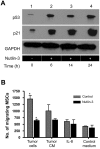
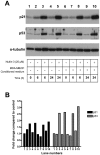
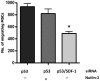
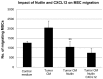
Mesenchymal stromal cells (MSCs) are multipotent fibroblast-like cells located in the bone marrow that localize to areas of tissue damage including wounds and solid tumors. Within the tumor microenvironment, MSCs adopt the phenotype of carcinoma-associated fibroblasts (CAFs) and stimulate tumor growth. Production of the chemokine CXCL12, also known as stromal cell-derived factor 1 (SDF-1), by MSCs is required for their in vitro migration in response to tumor cells and has also been implicated in stimulation of tumor growth. The tumor suppressor p53 regulates cellular migration, CXCL12 production and the promotion of tumor growth by carcinoma-associated fibroblasts (CAFs). We investigated the role of p53 in MSC migration to tumors. P53 inhibits the migration of MSCs in response to tumor cells in conjunction with a decrease in CXCL12 transcription. Conversely, decreased p53 activity leads to enhanced MSC migration. Interestingly, increased p53 activity inhibits MSC migration even in the context of high concentrations of exogenous CXCL12. These data show that stromal p53 status impacts the recruitment of MSCs to solid tumors through both regulation of CXCL12 production as well as other mechanisms. Stromal p53 may influence other important aspects of tumor biology such as tumor growth and metastasis through mechanisms distinct from CXCL12.
Figures




References
-
- Studeny M, Marini FC, Dembinski JL, Zompetta C, Cabreira-Hansen M, Bekele BN, Champlin RE, Andreeff M. Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J Natl Cancer Inst. 2004;96:1593–1603. - PubMed
-
- Studeny M, Marini FC, Champlin RE, Zompetta C, Fidler IJ, Andreeff M. Bone marrow-derived mesenchymal stem cells as vehicles for interferon-beta delivery into tumors. Cancer Res. 2002;62:3603–3608. - PubMed
-
- Shen FH, Visger JM, Balian G, Hurwitz SR, Diduch DR. Systemically administered mesenchymal stromal cells transduced with insulin-like growth factor-I localize to a fracture site and potentiate healing. J Orthop Trauma. 2002;16:651–659. - PubMed
-
- Rochefort GY, Delorme B, Lopez A, Herault O, Bonnet P, Charbord P, Eder V, Domenech J. Multipotential mesenchymal stem cells are mobilized into peripheral blood by hypoxia. Stem Cells. 2006;24:2202–2208. - PubMed
-
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

