Popeye domain containing 1 (Popdc1/Bves) is a caveolae-associated protein involved in ischemia tolerance
- PMID: 24066022
- PMCID: PMC3774711
- DOI: 10.1371/journal.pone.0071100
Popeye domain containing 1 (Popdc1/Bves) is a caveolae-associated protein involved in ischemia tolerance
Abstract
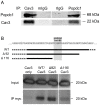
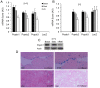
Popeye domain containing1 (Popdc1), also named Bves, is an evolutionary conserved membrane protein. Despite its high expression level in the heart little is known about its membrane localization and cardiac functions. The study examined the hypothesis that Popdc1 might be associated with the caveolae and play a role in myocardial ischemia tolerance. To address these issues, we analyzed hearts and cardiomyocytes of wild type and Popdc1-null mice. Immunoconfocal microscopy revealed co-localization of Popdc1 with caveolin3 in the sarcolemma, intercalated discs and T-tubules and with costameric vinculin. Popdc1 was co-immunoprecipitated with caveolin3 from cardiomyocytes and from transfected COS7 cells and was co-sedimented with caveolin3 in equilibrium density gradients. Caveolae disruption by methyl-β-cyclodextrin or by ischemia/reperfusion (I/R) abolished the cellular co-localization of Popdc1 with caveolin3 and modified their density co-sedimentation. The caveolin3-rich fractions of Popdc1-null hearts redistributed to fractions of lower buoyant density. Electron microscopy showed a statistically significant 70% reduction in caveolae number and a 12% increase in the average diameter of the remaining caveolae in the mutant hearts. In accordance with these changes, Popdc1-null cardiomyocytes displayed impaired [Ca(+2)]i transients, increased vulnerability to oxidative stress and no pharmacologic preconditioning. In addition, induction of I/R injury to Langendorff-perfused hearts indicated a significantly lower functional recovery in the mutant compared with wild type hearts while their infarct size was larger. No improvement in functional recovery was observed in Popdc1-null hearts following ischemic preconditioning. The results indicate that Popdc1 is a caveolae-associated protein important for the preservation of caveolae structural and functional integrity and for heart protection.
Conflict of interest statement
Figures







References
-
- Reese DE, Zavaljevski M, Streiff NL, Bader D (1999) bves:A Novel Gene Expressed during Coronary Blood Vessel Development. Dev Biol 209: 159–171. - PubMed
-
- Andree B, Hillemann T, Kessler-Icekson G, Schmitt-John T, Jockusch H, et al. (2000) Isolation and characterization of the novel popeye gene family expressed in skeletal muscle and heart. Dev Biol 223: 371–382. - PubMed
-
- Knight RF, Bader DM, Backstrom JR (2003) Membrane topology of Bves/Pop1A, a cell adhesion molecule that displays dynamic changes in cellular distribution during development. J Biol Chem 278: 32872–32879. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

