Cannabinoid transmission in the prelimbic cortex bidirectionally controls opiate reward and aversion signaling through dissociable kappa versus μ-opiate receptor dependent mechanisms
- PMID: 24068830
- PMCID: PMC6618460
- DOI: 10.1523/JNEUROSCI.1686-13.2013
Cannabinoid transmission in the prelimbic cortex bidirectionally controls opiate reward and aversion signaling through dissociable kappa versus μ-opiate receptor dependent mechanisms
Abstract
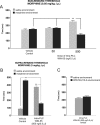
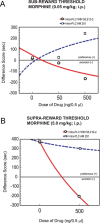
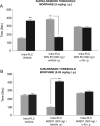
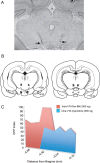
Cannabinoid, dopamine (DA), and opiate receptor pathways play integrative roles in emotional learning, associative memory, and sensory perception. Modulation of cannabinoid CB1 receptor transmission within the medial prefrontal cortex (mPFC) regulates the emotional valence of both rewarding and aversive experiences. Furthermore, CB1 receptor substrates functionally interact with opiate-related motivational processing circuits, particularly in the context of reward-related learning and memory. Considerable evidence demonstrates functional interactions between CB1 and DA signaling pathways during the processing of motivationally salient information. However, the role of mPFC CB1 receptor transmission in the modulation of behavioral opiate-reward processing is not currently known. Using an unbiased conditioned place preference paradigm with rats, we examined the role of intra-mPFC CB1 transmission during opiate reward learning. We report that activation or inhibition of CB1 transmission within the prelimbic cortical (PLC) division of the mPFC bidirectionally regulates the motivational valence of opiates; whereas CB1 activation switched morphine reward signaling into an aversive stimulus, blockade of CB1 transmission potentiated the rewarding properties of normally sub-reward threshold conditioning doses of morphine. Both of these effects were dependent upon DA transmission as systemic blockade of DAergic transmission prevented CB1-dependent modulation of morphine reward and aversion behaviors. We further report that CB1-mediated intra-PLC opiate motivational signaling is mediated through a μ-opiate receptor-dependent reward pathway, or a κ-opiate receptor-dependent aversion pathway, directly within the ventral tegmental area. Our results provide evidence for a novel CB1-mediated motivational valence switching mechanism within the PLC, controlling dissociable subcortical reward and aversion pathways.
Figures



References
-
- Auclair N, Otani S, Soubrie P, Crepel F. Cannabinoids modulate synaptic strength and plasticity at glutamatergic synapses of rat prefrontal cortex pyramidal neurons. J Neurophysiol. 2000;83:3287–3293. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
