Maturation of calcium-dependent GABA, glycine, and glutamate release in the glycinergic MNTB-LSO pathway
- PMID: 24069436
- PMCID: PMC3777950
- DOI: 10.1371/journal.pone.0075688
Maturation of calcium-dependent GABA, glycine, and glutamate release in the glycinergic MNTB-LSO pathway
Abstract
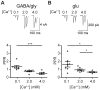
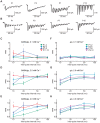
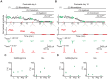
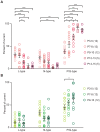
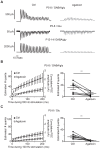
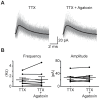
The medial nucleus of the trapezoid body (MNTB) is a key nucleus in high-fidelity temporal processing that underlies sound localization in the auditory brainstem. While the glycinergic principal cells of the MNTB project to all primary nuclei of the superior olive, during development the projection from MNTB to the lateral superior olive (LSO) is of interest because this immature inhibitory projection is known to undergo tonotopic refinement during an early postnatal period, and because during this period individual MNTB terminals in the LSO transiently release glycine GABA and glutamate. Developmental changes in calcium-dependent release are understood to be required to allow various auditory nuclei to follow high frequency activity; however, little is known about maturation of calcium-dependent release in the MNTB-LSO pathway, which has been presumed to have less stringent requirements for high-fidelity temporal following. In acute brainstem slices of rats age postnatal day 1 to 15 we recorded whole-cell responses in LSO principal neurons to electrical stimulation in the MNTB in order to measure sensitivity to external calcium, the contribution of different voltage-gated calcium channel subtypes to vesicular release, and the maturation of these measures for both GABA/glycine and glutamate transmission. Our results establish that release of glutamate at MNTB-LSO synapses is calcium-dependent. Whereas no significant developmental changes were evident for glutamate release, GABA/glycine release underwent substantial changes over the first two postnatal weeks: soon after birth L-type, N-type, and P/Q-type voltage-gated calcium channels (VGCCs) together mediated release, but after hearing onset P/Q-type VGCCs predominated. Blockade of P/Q-type VGCCs reduced the estimated quantal number for GABA/gly and glutamate transmission at P5-8 and the frequency of evoked miniature glycinergic events at P12-15, without apparent effects on spontaneous release of neurotransmitter, supporting a model in which P/Q-type VGCCs are required for mature synchronous synaptic transmission, but not for spontaneous vesicle release.
Conflict of interest statement
Figures

References
-
- Boudreau JC, Tsuchitani C (1968) Binaural interaction in the cat superior olive S segment. J Neurophysiol 31: 442–454. - PubMed
-
- Caird D, Klinke R (1983) Processing of binaural stimuli by cat superior olivary complex neurons. Exp Brain Res 52: 385–399. - PubMed
-
- Cant NB, Casseday JH (1986) Projections from the anteroventral cochlear nucleus to the lateral and medial superior olivary nuclei. J Comp Neurol 247: 457–476. - PubMed
-
- Kim G, Kandler K (2003) Elimination and strengthening of glycinergic/GABAergic connections during tonotopic map formation. Nat Neurosci 6: 282–290. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

