Ketogenic diet improves motor performance but not cognition in two mouse models of Alzheimer's pathology
- PMID: 24069439
- PMCID: PMC3771931
- DOI: 10.1371/journal.pone.0075713
Ketogenic diet improves motor performance but not cognition in two mouse models of Alzheimer's pathology
Abstract
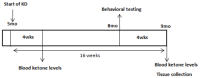
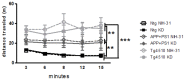
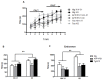
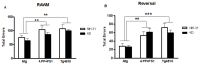
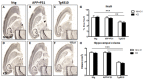
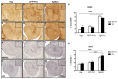
Dietary manipulations are increasingly viewed as possible approaches to treating neurodegenerative diseases. Previous studies suggest that Alzheimer's disease (AD) patients present an energy imbalance with brain hypometabolism and mitochondrial deficits. Ketogenic diets (KDs), widely investigated in the treatment and prevention of seizures, have been suggested to bypass metabolic deficits present in AD brain by providing ketone bodies as an alternative fuel to neurons. We investigated the effects of a ketogenic diet in two transgenic mouse lines. Five months old APP/PS1 (a model of amyloid deposition) and Tg4510 (a model of tau deposition) mice were offered either a ketogenic or a control (NIH-31) diet for 3 months. Body weight and food intake were monitored throughout the experiment, and blood was collected at 4 weeks and 4 months for ketone and glucose assessments. Both lines of transgenic mice weighed less than nontransgenic mice, yet, surprisingly, had elevated food intake. The ketogenic diet did not affect these differences in body weight or food consumption. Behavioral testing during the last two weeks of treatment found that mice offered KD performed significantly better on the rotarod compared to mice on the control diet independent of genotype. In the open field test, both transgenic mouse lines presented increased locomotor activity compared to nontransgenic, age-matched controls, and this effect was not influenced by KD. The radial arm water maze identified learning deficits in both transgenic lines with no significant differences between diets. Tissue measures of amyloid, tau, astroglial and microglial markers in transgenic lines showed no differences between animals fed the control or the ketogenic diet. These data suggest that ketogenic diets may play an important role in enhancing motor performance in mice, but have minimal impact on the phenotype of murine models of amyloid or tau deposition.
Conflict of interest statement
Figures
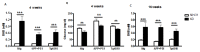

Similar articles
-
Partial rescue of memory deficits induced by calorie restriction in a mouse model of tau deposition.Behav Brain Res. 2014 Sep 1;271:79-88. doi: 10.1016/j.bbr.2014.06.001. Epub 2014 Jun 9. Behav Brain Res. 2014. PMID: 24925454
-
A ketone ester diet exhibits anxiolytic and cognition-sparing properties, and lessens amyloid and tau pathologies in a mouse model of Alzheimer's disease.Neurobiol Aging. 2013 Jun;34(6):1530-9. doi: 10.1016/j.neurobiolaging.2012.11.023. Epub 2012 Dec 29. Neurobiol Aging. 2013. PMID: 23276384 Free PMC article.
-
Ketogenic diet alleviates brain iron deposition and cognitive dysfunction via Nrf2-mediated ferroptosis pathway in APP/PS1 mouse.Brain Res. 2023 Aug 1;1812:148404. doi: 10.1016/j.brainres.2023.148404. Epub 2023 May 8. Brain Res. 2023. PMID: 37164173
-
Ketogenic Diet in Alzheimer's Disease.Int J Mol Sci. 2019 Aug 9;20(16):3892. doi: 10.3390/ijms20163892. Int J Mol Sci. 2019. PMID: 31405021 Free PMC article. Review.
-
Effects of CX3CR1 and Fractalkine Chemokines in Amyloid Beta Clearance and p-Tau Accumulation in Alzheimer's Disease (AD) Rodent Models: Is Fractalkine a Systemic Biomarker for AD?Curr Alzheimer Res. 2016;13(4):403-12. doi: 10.2174/1567205013666151116125714. Curr Alzheimer Res. 2016. PMID: 26567742 Review.
Cited by
-
The effect of ketogenic diet on behaviors and synaptic functions of naive mice.Brain Behav. 2019 Apr;9(4):e01246. doi: 10.1002/brb3.1246. Epub 2019 Mar 7. Brain Behav. 2019. PMID: 30848079 Free PMC article.
-
Ketone-Based Metabolic Therapy: Is Increased NAD+ a Primary Mechanism?Front Mol Neurosci. 2017 Nov 14;10:377. doi: 10.3389/fnmol.2017.00377. eCollection 2017. Front Mol Neurosci. 2017. PMID: 29184484 Free PMC article.
-
A ketogenic diet differentially affects neuron and astrocyte transcription.J Neurochem. 2021 Jun;157(6):1930-1945. doi: 10.1111/jnc.15313. Epub 2021 Feb 23. J Neurochem. 2021. PMID: 33539571 Free PMC article.
-
Gene Expression Signatures of Immaturity, Decreased pH, and Neural Hyperexcitation in the Hippocampus of Alzheimer's Disease Model Mice.Neuropsychopharmacol Rep. 2025 Mar;45(1):e70001. doi: 10.1002/npr2.70001. Neuropsychopharmacol Rep. 2025. PMID: 39907034 Free PMC article.
-
RanBP9 at the intersection between cofilin and Aβ pathologies: rescue of neurodegenerative changes by RanBP9 reduction.Cell Death Dis. 2015 Mar 5;6(3):1676. doi: 10.1038/cddis.2015.37. Cell Death Dis. 2015. PMID: 25741591 Free PMC article.
References
-
- Foster NL, Chase TN, Fedio P, Patronas NJ, Brooks RA et al. (1983) Alzheimer’s disease: focal cortical changes shown by positron emission tomography. Neurology 33: 961-965. doi:10.1212/WNL.33.8.961. PubMed: 6603596. - DOI - PubMed
-
- Costantini LC, Barr LJ, Vogel JL, Henderson ST (2008) Hypometabolism as a therapeutic target in Alzheimer’s disease. BMC Neurosci 9 Suppl 2: S16. doi:10.1186/1471-2202-9-S2-S16. PubMed: 19090989. - DOI - PMC - PubMed
-
- Diehl J, Grimmer T, Drzezga A, Riemenschneider M, Förstl H et al. (2004) Cerebral metabolic patterns at early stages of frontotemporal dementia and semantic dementia. A PET study. Neurobiol Aging 25: 1051-1056. doi:10.1016/j.neurobiolaging.2003.10.007. PubMed: 15212830. - DOI - PubMed
-
- Diehl-Schmid J, Grimmer T, Drzezga A, Bornschein S, Riemenschneider M et al. (2007) Decline of cerebral glucose metabolism in frontotemporal dementia: a longitudinal 18F-FDG-PET-study. Neurobiol Aging 28: 42-50. doi:10.1016/j.neurobiolaging.2005.11.002. PubMed: 16448722. - DOI - PubMed
-
- Blass JP, Sheu RK, Gibson GE (2000) Inherent abnormalities in energy metabolism in Alzheimer disease. Interaction with cerebrovascular compromise. Ann N Y Acad Sci 903: 204-221. doi:10.1111/j.1749-6632.2000.tb06370.x. PubMed: 10818509. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

