Nanoelectronics-biology frontier: From nanoscopic probes for action potential recording in live cells to three-dimensional cyborg tissues
- PMID: 24073014
- PMCID: PMC3781175
- DOI: 10.1016/j.nantod.2013.05.001
Nanoelectronics-biology frontier: From nanoscopic probes for action potential recording in live cells to three-dimensional cyborg tissues
Abstract
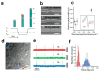
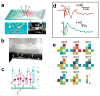
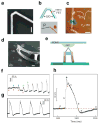
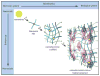
Semiconductor nanowires configured as the active channels of field-effect transistors (FETs) have been used as detectors for high-resolution electrical recording from single live cells, cell networks, tissues and organs. Extracellular measurements with substrate supported silicon nanowire (SiNW) FETs, which have projected active areas orders of magnitude smaller than conventional microfabricated multielectrode arrays (MEAs) and planar FETs, recorded action potential and field potential signals with high signal-to-noise ratio and temporal resolution from cultured neurons, cultured cardiomyocytes, acute brain slices and whole animal hearts. Measurements made with modulation-doped nanoscale active channel SiNW FETs demonstrate that signals recorded from cardiomyocytes are highly localized and have improved time resolution compared to larger planar detectors. In addition, several novel three-dimensional (3D) transistor probes, which were realized using advanced nanowire synthesis methods, have been implemented for intracellular recording. These novel probes include (i) flexible 3D kinked nanowire FETs, (ii) branched intracellular nanotube SiNW FETs, and (iii) active silicon nanotube FETs. Following phospholipid modification of the probes to mimic the cell membrane, the kinked nanowire, branched intracellular nanotube and active silicon nanotube FET probes recorded full-amplitude intracellular action potentials from spontaneously firing cardiomyocytes. Moreover, these probes demonstrated the capability of reversible, stable, and long-term intracellular recording, thus indicating the minimal invasiveness of the new nanoscale structures and suggesting biomimetic internalization via the phospholipid modification. Simultaneous, multi-site intracellular recording from both single cells and cell networks were also readily achieved by interfacing independently addressable nanoprobe devices with cells. Finally, electronic and biological systems have been seamlessly merged in 3D for the first time using macroporous nanoelectronic scaffolds that are analogous to synthetic tissue scaffold and the extracellular matrix in tissue. Free-standing 3D nanoelectronic scaffolds were cultured with neurons, cardiomyocytes and smooth muscle cells to yield electronically-innervated synthetic or 'cyborg' tissues. Measurements demonstrate that innervated tissues exhibit similar cell viability as with conventional tissue scaffolds, and importantly, demonstrate that the real-time response to drugs and pH changes can be mapped in 3D through the tissues. These results open up a new field of research, wherein nanoelectronics are merged with biological systems in 3D thereby providing broad opportunities, ranging from a nanoelectronic/tissue platform for real-time pharmacological screening in 3D to implantable 'cyborg' tissues enabling closed-loop monitoring and treatment of diseases. Furthermore, the capability of high density scale-up of the above extra- and intracellular nanoscopic probes for action potential recording provide important tools for large-scale high spatio-temporal resolution electrical neural activity mapping in both 2D and 3D, which promises to have a profound impact on many research areas, including the mapping of activity within the brain.
Keywords: bioelectronics; brain activity mapping; cellular and subcellular resolution; field-effect transistor; flexible electronics; graphene; macroporous 3D electronics; nanodevice; nanowire; synthetic tissue.
Figures







References
-
- Hille B. Ion Channels of Excitable Membranes. 3. Sinauer Associates Inc; Sunderland: 2001.
-
- Dunlop J, Bowlby M, Peri R, Vasilyev D, Arias R. Nature Rev Drug Discov. 2008;7:358–368. - PubMed
-
- Meyer T, Boven KH, Gunther E, Fejtl M. Drug Safety. 2004;27:763–772. - PubMed
-
- Dhein S, Mohr FW, Delmar M. Practical Methods in Cardiovascular Research. Springer-Verlag; Berlin: 2005. pp. 215–453.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
