The Taz1p transacylase is imported and sorted into the outer mitochondrial membrane via a membrane anchor domain
- PMID: 24078306
- PMCID: PMC3889572
- DOI: 10.1128/EC.00237-13
The Taz1p transacylase is imported and sorted into the outer mitochondrial membrane via a membrane anchor domain
Abstract
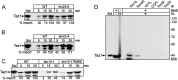
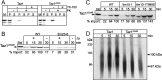
Mutations in the mitochondrial transacylase tafazzin, Taz1p, in Saccharomyces cerevisiae cause Barth syndrome, a disease of defective cardiolipin remodeling. Taz1p is an interfacial membrane protein that localizes to both the outer and inner membranes, lining the intermembrane space. Pathogenic point mutations in Taz1p that alter import and membrane insertion result in accumulation of monolysocardiolipin. In this study, we used yeast as a model to investigate the biogenesis of Taz1p. We show that to achieve this unique topology in mitochondria, Taz1p follows a novel import pathway in which it crosses the outer membrane via the translocase of the outer membrane and then uses the Tim9p-Tim10p complex of the intermembrane space to insert into the mitochondrial outer membrane. Taz1p is then transported to membranes of an intermediate density to reach a location in the inner membrane. Moreover, a pathogenic mutation within the membrane anchor (V224R) alters Taz1p import so that it bypasses the Tim9p-Tim10p complex and interacts with the translocase of the inner membrane, TIM23, to reach the matrix. Critical targeting information for Taz1p resides in the membrane anchor and flanking sequences, which are often mutated in Barth syndrome patients. These studies suggest that altering the mitochondrial import pathway of Taz1p may be important in understanding the molecular basis of Barth syndrome.
Figures




Similar articles
-
Mitochondrial mislocalization and altered assembly of a cluster of Barth syndrome mutant tafazzins.J Cell Biol. 2006 Jul 31;174(3):379-90. doi: 10.1083/jcb.200605043. J Cell Biol. 2006. PMID: 16880272 Free PMC article.
-
Barth syndrome mutations that cause tafazzin complex lability.J Cell Biol. 2011 Feb 7;192(3):447-62. doi: 10.1083/jcb.201008177. J Cell Biol. 2011. PMID: 21300850 Free PMC article.
-
Deacylation on the matrix side of the mitochondrial inner membrane regulates cardiolipin remodeling.Mol Biol Cell. 2013 Jun;24(12):2008-20. doi: 10.1091/mbc.E13-03-0121. Epub 2013 May 1. Mol Biol Cell. 2013. PMID: 23637464 Free PMC article.
-
Cardiolipin function in the yeast S. cerevisiae and the lessons learned for Barth syndrome.J Inherit Metab Dis. 2022 Jan;45(1):60-71. doi: 10.1002/jimd.12447. Epub 2021 Oct 19. J Inherit Metab Dis. 2022. PMID: 34626131 Free PMC article. Review.
-
Experimental models of Barth syndrome.J Inherit Metab Dis. 2022 Jan;45(1):72-81. doi: 10.1002/jimd.12423. Epub 2021 Aug 15. J Inherit Metab Dis. 2022. PMID: 34370877 Free PMC article. Review.
Cited by
-
TAZ encodes tafazzin, a transacylase essential for cardiolipin formation and central to the etiology of Barth syndrome.Gene. 2020 Feb 5;726:144148. doi: 10.1016/j.gene.2019.144148. Epub 2019 Oct 21. Gene. 2020. PMID: 31647997 Free PMC article. Review.
-
Identification of novel mitochondrial localization signals in human Tafazzin, the cause of the inherited cardiomyopathic disorder Barth syndrome.J Mol Cell Cardiol. 2018 Jan;114:83-92. doi: 10.1016/j.yjmcc.2017.11.005. Epub 2017 Nov 10. J Mol Cell Cardiol. 2018. PMID: 29129703 Free PMC article.
-
Single Cell Transcriptomic Analysis in a Mouse Model of Barth Syndrome Reveals Cell-Specific Alterations in Gene Expression and Intercellular Communication.Int J Mol Sci. 2023 Jul 18;24(14):11594. doi: 10.3390/ijms241411594. Int J Mol Sci. 2023. PMID: 37511352 Free PMC article.
-
Role of Tafazzin in Mitochondrial Function, Development and Disease.J Dev Biol. 2020 May 23;8(2):10. doi: 10.3390/jdb8020010. J Dev Biol. 2020. PMID: 32456129 Free PMC article. Review.
-
Aim24 and MICOS modulate respiratory function, tafazzin-related cardiolipin modification and mitochondrial architecture.Elife. 2014 Jan 1;3:e01684. doi: 10.7554/eLife.01684. Elife. 2014. PMID: 24714493 Free PMC article.
References
-
- Barth PG, Valianpour F, Bowen VM, Lam J, Duran M, Vaz FM, Wanders RJ. 2004. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome): an update. Am. J. Med. Genet. A 126A:349–354 - PubMed
-
- Li G, Chen S, Thompson MN, Greenberg ML. 2007. New insights into the regulation of cardiolipin biosynthesis in yeast: implications for Barth syndrome. Biochim. Biophys. Acta 1771:432–441 - PubMed
-
- Barth PG, Wanders RJ, Vreken P, Janssen EA, Lam J, Baas F. 1999. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome). J. Inherit Metab. Dis. 22:555–567 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

