Sublethal heat shock induces premature senescence rather than apoptosis in human mesenchymal stem cells
- PMID: 24078383
- PMCID: PMC3982025
- DOI: 10.1007/s12192-013-0463-6
Sublethal heat shock induces premature senescence rather than apoptosis in human mesenchymal stem cells
Abstract
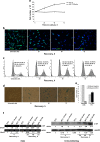
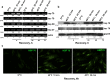
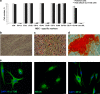
Stem cells in adult organism are responsible for cell turnover and tissue regeneration. The study of stem cell stress response contributes to our knowledge on the mechanisms of damaged tissue repair. Previously, we demonstrated that sublethal heat shock (HS) induced apoptosis in human embryonic stem cells. This study aimed to investigate HS response of human adult stem cells. Human mesenchymal stem cells (MSCs) cultivated in vitro were challenged with sublethal HS. It was found that sublethal HS did not affect the cell viability assessed by annexin V/propidium staining. However, MSCs subjected to severe HS exhibited features of stress-induced premature senescence (SIPS): irreversible cell cycle arrest, altered morphology, increased expression of senescence-associated β-galactosidase (SA-β-gal) activity, and induction of cyclin-dependent kinase inhibitor p21 protein. High level of Hsp70 accumulation induced by sublethal HS did not return to the basal level, at least, after 72 h of the cell recovery when most cells exhibited SIPS hallmarks. MSCs survived sublethal HS, and resumed proliferation sustained the properties of parental MSCs: diploid karyotype, replicative senescence, expression of the cell surface markers, and capacity for multilineage differentiation. Our results showed for the first time that in human MSCs, sublethal HS induced premature senescence rather than apoptosis or necrosis. MSC progeny that survived sublethal HS manifested stem cell properties of the parental cells: limited replicative life span and multilineage capacity.
Figures


References
-
- Alekseenko LL, Zemelko VI, Zenin VV, Pugovkina NA, Kozhukharova IV, Kovaleva ZV, Grinchuk TM, Fridlyanskaya II, Nikolsky NN. Heat shock induces apoptosis in human embryonic stem cells but a premature senescence phenotype in their differentiated progeny. Cell Cycle. 2012;11:3260–3269. doi: 10.4161/cc.21595. - DOI - PMC - PubMed
-
- Bladier C, Wolvetang EJ, Hutchinson P, de Haan JB, Kola I. Response of a primary human fibroblast cell line to H2O2: senescence-like growth arrest or apoptosis? Cell Growth Differ. 1997;8:589–598. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

