Nitric oxide, S-nitrosation, and endothelial permeability
- PMID: 24078390
- PMCID: PMC4004350
- DOI: 10.1002/iub.1204
Nitric oxide, S-nitrosation, and endothelial permeability
Abstract
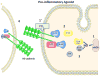
S-Nitrosation is rapidly emerging as a regulatory mechanism in vascular biology, with particular importance in the onset of hyperpermeability induced by pro-inflammatory agents. This review focuses on the role of endothelial nitric oxide synthase (eNOS)-derived nitric oxide (NO) in regulating S-Nitrosation of adherens junction proteins. We discuss evidence for translocation of eNOS, via caveolae, to the cytosol and analyze the significance of eNOS location for S-Nitrosation and onset of endothelial hyperpermeability to macromolecules.
Keywords: S-Nitrosation; VE-cadherin; adherens junction proteins; endothelial nitric oxide synthase; endothelial permeability; endothelium; microvascular permeability; nitric oxide; p120-catenin; regulation of inflammatory hyperpermeability.
© 2013 International Union of Biochemistry and Molecular Biology.
Figures


References
-
- Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288:373–376. - PubMed
-
- Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987;327:524–526. - PubMed
-
- Huang Q, Yuan Y. Interaction of pkc and nos in signal transduction of microvascular hyperpermeability. Am J Physiol. 1997;273:H2442–2451. - PubMed
-
- Varma S, Breslin JW, Lal BK, Pappas PJ, Hobson RW, 2nd, Duran WN. P42/44mapk regulates baseline permeability and cgmp-induced hyperpermeability in endothelial cells. Microvasc Res. 2002;63:172–178. - PubMed
-
- Kramer GC, Harms BA, Bodai BI, Demling RH, Renkin EM. Mechanisms for redistribution of plasma protein following acute protein depletion. Am J Physiol. 1982;243:H803–809. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

