Safety, tolerability, and pharmacokinetics of ribavirin in hepatitis C virus-infected patients with various degrees of renal impairment
- PMID: 24080649
- PMCID: PMC3837852
- DOI: 10.1128/AAC.00608-13
Safety, tolerability, and pharmacokinetics of ribavirin in hepatitis C virus-infected patients with various degrees of renal impairment
Abstract
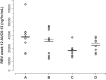
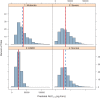
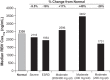
Ribavirin (RBV) is an integral part of standard-of-care hepatitis C virus (HCV) treatments and many future regimens under investigation. The pharmacokinetics (PK), safety, and tolerability of RBV in chronically HCV-infected patients with renal impairment are not well defined and were the focus of an open-label PK study in HCV-infected patients receiving RBV plus pegylated interferon. Serial RBV plasma samples were collected over 12 h on day 1 of weeks 1 and 12 from patients with moderate renal impairment (creatinine clearance [CLCR], 30 to 50 ml/min; RBV, 600 mg daily), severe renal impairment (CLCR, <30 ml/min; RBV, 400 mg daily), end-stage renal disease (ESRD) (RBV, 200 mg daily), or normal renal function (CLCR, >80 ml/min; RBV, 800 to 1,200 mg daily). Of the 44 patients, 9 had moderately impaired renal function, 10 had severely impaired renal function, 13 had ESRD, and 12 had normal renal function. The RBV dose was reduced because of adverse events (AEs) in 71% and 53% of severe and moderate renal impairment groups, respectively. Despite this modification, patients with moderate and severe impairment had 12-hour (area under the concentration-time curve from 0 to 12 h [AUC0-12]) values 36% (38,452 ng · h/ml) and 25% (35,101 ng · h/ml) higher, respectively, than those with normal renal function (28,192 ng · h/ml). Patients with ESRD tolerated a 200-mg daily dose, and AUC0-12 was 20% lower (22,629 ng · h/ml) than in patients with normal renal function. PK modeling and simulation (M&S) indicated that doses of 200 mg or 400 mg alternating daily for patients with moderate renal impairment and 200 mg daily for patients with severe renal impairment were the most appropriate dose regimens in these patients.
Figures

References
-
- Chak E, Talal AH, Sherman KE, Schiff ER, Saab S. 2011. Hepatitis C virus infection in USA: an estimate of true prevalence. Liver Int. 31:1090–1101 - PubMed
-
- Lavanchy D. 2009. The global burden of hepatitis C. Liver Int. 29(Suppl 1):74–81 - PubMed
-
- Butt AA, Umbleja T, Andersen JW, Chung RT, Sherman KE. 2011. The incidence, predictors and management of anaemia and its association with virological response in HCV/HIV coinfected persons treated with long-term pegylated interferon alfa 2a and ribavirin. Aliment. Pharmacol. Ther. 33:1234–1244 - PMC - PubMed
-
- Perico N, Cattaneo D, Bikbov B, Remuzzi G. 2009. Hepatitis C infection and chronic renal diseases. Clin. J. Am. Soc. Nephrol. 4:207–220 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

