High-throughput profiling of anti-glycan humoral responses to SIV vaccination and challenge
- PMID: 24086502
- PMCID: PMC3781036
- DOI: 10.1371/journal.pone.0075302
High-throughput profiling of anti-glycan humoral responses to SIV vaccination and challenge
Erratum in
- PLoS One. 2013;8(12). doi:10.1371/annotation/fd9f4a65-3f27-46cd-8ed0-6c9b8ba7f83b. Damberg, Thorsten [corrected to Demberg, Thorsten]
Abstract
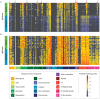
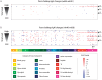
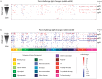
Recent progress toward an HIV vaccine highlights both the potential of vaccines to end the AIDS pandemic and the need to boost efficacy by incorporating additional vaccine strategies. Although many aspects of the immune response can contribute to vaccine efficacy, the key factors have not been defined fully yet. A particular area that may yield new insights is anti-glycan immune responses, such as those against the glycan shield that HIV uses to evade the immune system. In this study, we used glycan microarray technology to evaluate anti-glycan antibody responses induced by SIV vaccination and infection in a non-human primate model of HIV infection. This comprehensive profiling of circulating anti-glycan antibodies found changes in anti-glycan antibody levels after both vaccination with the Ad5hr-SIV vaccine and SIV infection. Notably, SIV infection produced generalized declines in anti-glycan IgM antibodies in a number of animals. Additionally, some infected animals generated antibodies to the Tn antigen, which is a cryptic tumor-associated antigen exposed by premature termination of O-linked glycans; however, the Ad5hr-SIV vaccine did not induce anti-Tn IgG antibodies. Overall, this study demonstrates the potential contributions that glycan microarrays can make for HIV vaccine development.
Conflict of interest statement
Figures


Similar articles
-
Early T Follicular Helper Cell Responses and Germinal Center Reactions Are Associated with Viremia Control in Immunized Rhesus Macaques.J Virol. 2019 Feb 5;93(4):e01687-18. doi: 10.1128/JVI.01687-18. Print 2019 Feb 15. J Virol. 2019. PMID: 30463978 Free PMC article.
-
Analysis of Complement-Mediated Lysis of Simian Immunodeficiency Virus (SIV) and SIV-Infected Cells Reveals Sex Differences in Vaccine-Induced Immune Responses in Rhesus Macaques.J Virol. 2018 Sep 12;92(19):e00721-18. doi: 10.1128/JVI.00721-18. Print 2018 Oct 1. J Virol. 2018. PMID: 30021899 Free PMC article.
-
Preexisting vaccinia virus immunity decreases SIV-specific cellular immunity but does not diminish humoral immunity and efficacy of a DNA/MVA vaccine.J Immunol. 2010 Dec 15;185(12):7262-73. doi: 10.4049/jimmunol.1000751. Epub 2010 Nov 12. J Immunol. 2010. PMID: 21076059 Free PMC article.
-
Anti-HIV and -SIV immunity in the vagina.Int Rev Immunol. 2003 Jan-Feb;22(1):65-76. doi: 10.1080/08830180305230. Int Rev Immunol. 2003. PMID: 12710504 Review.
-
Dissecting the antibody-OME: past, present, and future.Curr Opin Immunol. 2020 Aug;65:89-96. doi: 10.1016/j.coi.2020.06.003. Epub 2020 Aug 2. Curr Opin Immunol. 2020. PMID: 32755751 Review.
Cited by
-
Mass Spectrometric and Glycan Microarray-Based Characterization of the Filarial Nematode Brugia malayi Glycome Reveals Anionic and Zwitterionic Glycan Antigens.Mol Cell Proteomics. 2022 May;21(5):100201. doi: 10.1016/j.mcpro.2022.100201. Epub 2022 Jan 20. Mol Cell Proteomics. 2022. PMID: 35065273 Free PMC article.
-
Cooperation between somatic mutation and germline-encoded residues enables antibody recognition of HIV-1 envelope glycans.PLoS Pathog. 2019 Dec 16;15(12):e1008165. doi: 10.1371/journal.ppat.1008165. eCollection 2019 Dec. PLoS Pathog. 2019. PMID: 31841553 Free PMC article.
-
Serum glycan-binding IgG antibodies in HIV-1 infection and during the development of broadly neutralizing responses.AIDS. 2017 Oct 23;31(16):2199-2209. doi: 10.1097/QAD.0000000000001643. AIDS. 2017. PMID: 28926408 Free PMC article.
-
Construction and Evaluation of a Novel MAP Immunoassay for 9 Nutrition-and-Health-Related Protein Markers Based on Multiplex Liquid Protein Chip Technique.Nutrients. 2023 Mar 21;15(6):1522. doi: 10.3390/nu15061522. Nutrients. 2023. PMID: 36986252 Free PMC article.
-
Serum antibody screening using glycan arrays.Chem Soc Rev. 2024 Mar 4;53(5):2603-2642. doi: 10.1039/d3cs00693j. Chem Soc Rev. 2024. PMID: 38305761 Free PMC article. Review.
References
-
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, et al. (2009) Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med 361: 2209–2220. - PubMed
-
- Wei X, Decker JM, Wang S, Hui H, Kappes JC, et al. (2003) Antibody neutralization and escape by HIV-1. Nature 422: 307–312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

