Influence of apolipoprotein A-I and apolipoprotein A-II availability on nascent HDL heterogeneity
- PMID: 24089247
- PMCID: PMC3826692
- DOI: 10.1194/jlr.M043109
Influence of apolipoprotein A-I and apolipoprotein A-II availability on nascent HDL heterogeneity
Abstract
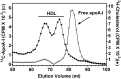
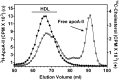
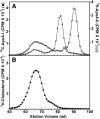
It is important to understand HDL heterogeneity because various subspecies possess different functionalities. To understand the origins of HDL heterogeneity arising from the existence of particles containing only apoA-I (LpA-I) and particles containing both apoA-I and apoA-II (LpA-I+A-II), we compared the abilities of both proteins to promote ABCA1-mediated efflux of cholesterol from HepG2 cells and form nascent HDL particles. When added separately, exogenous apoA-I and apoA-II were equally effective in promoting cholesterol efflux, although the resultant LpA-I and LpA-II particles had different sizes. When apoA-I and apoA-II were mixed together at initial molar ratios ranging from 1:1 to 16:1 to generate nascent LpA-I+A-II HDL particles, the particle size distribution altered, and the two proteins were incorporated into the nascent HDL in proportion to their initial ratio. Both proteins formed nascent HDL particles with equal efficiency, and the relative amounts of apoA-I and apoA-II incorporation were driven by mass action. The ratio of lipid-free apoA-I and apoA-II available at the surface of ABCA1-expressing cells is a major factor in determining the contents of these proteins in nascent HDL. Manipulation of this ratio provides a means of altering the relative distribution of LpA-I and LpA-I+A-II HDL particles.
Keywords: ATP binding cassette A1; cholesterol; lipoprotein; phospholipid.
Figures



Similar articles
-
Apolipoprotein A-II alters the proteome of human lipoproteins and enhances cholesterol efflux from ABCA1.J Lipid Res. 2017 Jul;58(7):1374-1385. doi: 10.1194/jlr.M075382. Epub 2017 May 5. J Lipid Res. 2017. PMID: 28476857 Free PMC article.
-
Influence of apolipoprotein (Apo) A-I structure on nascent high density lipoprotein (HDL) particle size distribution.J Biol Chem. 2010 Oct 15;285(42):31965-73. doi: 10.1074/jbc.M110.126292. Epub 2010 Aug 2. J Biol Chem. 2010. PMID: 20679346 Free PMC article.
-
Molecular and cellular physiology of apolipoprotein A-I lipidation by the ATP-binding cassette transporter A1 (ABCA1).J Biol Chem. 2004 Feb 27;279(9):7384-94. doi: 10.1074/jbc.M306963200. Epub 2003 Dec 4. J Biol Chem. 2004. PMID: 14660648
-
ATP-binding cassette transporter AI and its role in HDL formation.Curr Opin Lipidol. 2005 Feb;16(1):19-25. doi: 10.1097/00041433-200502000-00005. Curr Opin Lipidol. 2005. PMID: 15650559 Review.
-
Role of apoA-I, ABCA1, LCAT, and SR-BI in the biogenesis of HDL.J Mol Med (Berl). 2006 Apr;84(4):276-94. doi: 10.1007/s00109-005-0030-4. Epub 2006 Feb 25. J Mol Med (Berl). 2006. PMID: 16501936 Review.
Cited by
-
Apolipoprotein A-II alters the proteome of human lipoproteins and enhances cholesterol efflux from ABCA1.J Lipid Res. 2017 Jul;58(7):1374-1385. doi: 10.1194/jlr.M075382. Epub 2017 May 5. J Lipid Res. 2017. PMID: 28476857 Free PMC article.
-
Hot spots in apolipoprotein A-II misfolding and amyloidosis in mice and men.FEBS Lett. 2014 Mar 18;588(6):845-50. doi: 10.1016/j.febslet.2014.01.066. Epub 2014 Feb 20. FEBS Lett. 2014. PMID: 24561203 Free PMC article.
References
-
- Lewis G. F., Rader D. J. 2005. New insights into the regulation of HDL metabolism and reverse cholesterol transport. Circ. Res. 96: 1221–1232 - PubMed
-
- Yancey P. G., Bortnick A. E., Kellner-Weibel G., De La Llera-Moya M., Phillips M. C., Rothblat G. H. 2003. Importance of different pathways of cellular cholesterol efflux. Arterioscler. Thromb. Vasc. Biol. 23: 712–719 - PubMed
-
- Curtiss L. K., Valenta D. T., Hime N. J., Rye K. A. 2006. What is so special about apolipoprotein AI in reverse cholesterol transport? Arterioscler. Thromb. Vasc. Biol. 26: 12–19 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

