Integration of left-right Pitx2 transcription and Wnt signaling drives asymmetric gut morphogenesis via Daam2
- PMID: 24091014
- PMCID: PMC3965270
- DOI: 10.1016/j.devcel.2013.07.019
Integration of left-right Pitx2 transcription and Wnt signaling drives asymmetric gut morphogenesis via Daam2
Abstract
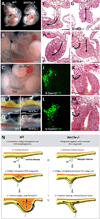
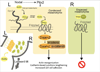
A critical aspect of gut morphogenesis is initiation of a leftward tilt, and failure to do so leads to gut malrotation and volvulus. The direction of tilt is specified by asymmetric cell behaviors within the dorsal mesentery (DM), which suspends the gut tube, and is downstream of Pitx2, the key transcription factor responsible for the transfer of left-right (L-R) information from early gastrulation to morphogenesis. Although Pitx2 is a master regulator of L-R organ development, its cellular targets that drive asymmetric morphogenesis are not known. Using laser microdissection and targeted gene misexpression in the chicken DM, we show that Pitx2-specific effectors mediate Wnt signaling to activate the formin Daam2, a key Wnt effector and itself a Pitx2 target, linking actin dynamics to cadherin-based junctions to ultimately generate asymmetric cell behaviors. Our work highlights how integration of two conserved cascades may be the ultimate force through which Pitx2 sculpts L-R organs.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures


Comment in
-
Your gut is right to turn left.Dev Cell. 2013 Sep 30;26(6):553-4. doi: 10.1016/j.devcel.2013.08.018. Dev Cell. 2013. PMID: 24091006 Free PMC article.
References
-
- Aspenstrom P, Richnau N, Johansson AS. The diaphanous-related formin DAAM1 collaborates with the Rho GTPases RhoA and Cdc42, CIP4 and Src in regulating cell morphogenesis and actin dynamics. Exp Cell Res. 2006;312:2180–2194. - PubMed
-
- Baeg GH, Lin X, Khare N, Baumgartner S, Perrimon N. Heparan sulfate proteoglycans are critical for the organization of the extracellular distribution of Wingless. Development. 2001;128:87–94. - PubMed
-
- Barolo S. Transgenic Wnt/TCF pathway reporters: all you need is Lef? Oncogene. 2006;25:7505–7511. - PubMed
-
- Biechele TL, Adams AM, Moon RT. Transcription-based reporters of Wnt/beta-catenin signaling. Cold Spring Harbor protocols. 2009;2009 pdb prot5223. - PubMed
-
- Brent AE, Schweitzer R, Tabin CJ. A somitic compartment of tendon progenitors. Cell. 2003;113:235–248. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

