Efficacy of deferred dosing of granulocyte colony-stimulating factor in autologous hematopoietic transplantation for multiple myeloma
- PMID: 24096822
- PMCID: PMC3915247
- DOI: 10.1038/bmt.2013.149
Efficacy of deferred dosing of granulocyte colony-stimulating factor in autologous hematopoietic transplantation for multiple myeloma
Abstract
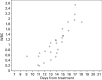
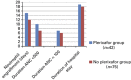
Routine administration of G-CSF following autologous hematopoietic SCT (ASCT) expedites ANC recovery and reduces hospitalization by 1-2 days; it has no impact on febrile neutropenia, infections, morbidity, mortality, event-free survival or OS. To determine whether delayed G-CSF dosage could result in equivalent ANC recovery and thereby improve cost effectiveness, we deferred the administration of G-CSF until WBC recovery had begun. A total of 117 patients with multiple myeloma received ASCT from January 2005 to September 2012. Of these, 52 were in the conventional dosing group (CGD) and received G-CSF from Day +7 for a median of five doses. In the deferred dosing group (DGD), 65 patients received G-CSF from median day 14 post transplant for a median of zero doses. There was no difference between groups in the incidence or duration of febrile neutropenia, duration of grade III mucositis, weight gain, rash, engraftment syndrome or early death (100 days). The DGD group had a significantly longer time to neutrophil engraftment than the CGD group (15 days vs 12 days; P<0.0001), a longer period of severe neutropenia (<100/μL; 8 days vs 6 days; P<0.0001), longer treatment with intravenous antibiotics (7 days vs 5 days; P=0.016) and longer hospital stay (19 days vs 17 days; P=<0.0001). Although the cost of G-CSF was lower in the DGD group (mean $308 vs $2467), the additional hospitalization raised the median total cost of ASCT in this group by 17%. There was, however, no adverse effect of deferred dosing on the rate of febrile neuropenic episodes or Day 100 survival, so that deferred dosing of G-CSF may be suitable for patients receiving ASCT as outpatients, for whom longer hospital stay would not be an offsetting cost.
Figures
References
-
- Klumpp TR, Mangan KF, Goldberg SL, Pearlman ES, Macdonald JS. Granulocyte colony-stimulating factor accelerates neutrophil engraftment following peripheral-blood stem-cell transplantation: a prospective, randomized trial. J Clin Oncol. 1995;13:1323–1327. - PubMed
-
- Samaras P, Blickenstorfer M, Siciliano RD, Haile SR, Buset EM, Petrausch U, et al. Pegfilgrastim reduces the length of hospitalization and the time to engraftment in multiple myeloma patients treated with melphalan 200 and auto-SCT compared with filgrastim. Ann Hematol. 2011;90:89–94. - PubMed
-
- Spitzer G, Adkins DR, Spencer V, Dunphy FR, Petruska PJ, Velasquez WS, et al. Randomized study of growth factors post-peripheral-blood stem-cell transplant: neutrophil recovery is improved with modest clinical benefit. J Clin Oncol. 1994;12:661–670. - PubMed
-
- Schmitz N, Ljungman P, Cordonnier C, Kampf C, Linkesch W, Alegre A, et al. Lenograstim after autologous peripheral blood progenitor cell transplantation: results of a double-blind, randomized trial. Bone Marrow Transplant. 2004;34:955–962. - PubMed
-
- McQuaker IG, Hunter AE, Pacey SHaynes AP, Iqbal A, Russell NH. Low-dose filgrastim significantly enhances neutrophil recovery following autologous peripheral-blood stem-cell transplantation in patients with lymphoproliferative disorders: evidence for clinical and economic benefit. J Clin Oncol. 1997;15:451–457. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical