Caprine abscess model of tulathromycin concentrations in interstitial fluid from tissue chambers inoculated with Corynebacterium pseudotuberculosis following subcutaneous or intrachamber administration
- PMID: 24100501
- PMCID: PMC3837882
- DOI: 10.1128/AAC.00936-13
Caprine abscess model of tulathromycin concentrations in interstitial fluid from tissue chambers inoculated with Corynebacterium pseudotuberculosis following subcutaneous or intrachamber administration
Abstract
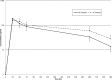
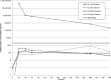
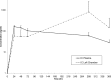
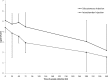
Corynebacterium pseudotuberculosis causes chronic, suppurative, abscessing conditions in livestock and humans. We used an in vivo model to evaluate antimicrobial efficacy for focal abscesses caused by C. pseudotuberculosis. Tissue chambers were surgically implanted in the subcutaneous tissues of the right and left paralumbar fossa of 12 goats to serve as a model for isolated, focal abscesses. For each goat, one tissue chamber was inoculated with C. pseudotuberculosis, while the contralateral chamber served as an uninoculated control. Six goats were administered a single dose of tulathromycin at 2.5 mg/kg of body weight subcutaneously, while the other six received the same dose by injection directly into the inoculated chambers. Our objective was to compare the effects and tulathromycin concentrations in interstitial fluid (IF) samples collected from C. pseudotuberculosis-infected and control chambers following subcutaneous or intrachamber injection of tulathromycin. In addition, the effects of tulathromycin on the quantity of C. pseudotuberculosis reisolated from inoculated chambers were assessed over time. Tulathromycin IF concentrations from C. pseudotuberculosis-infected and control tissue chambers were similar to those in plasma following subcutaneous administration. Following intrachamber administration, tulathromycin IF concentrations in infected chambers were continuously above the MIC for the C. pseudotuberculosis isolate for 15 days. There were no significant differences for plasma area under the curve and elimination half-lives between subcutaneous and intrachamber administration. Six of the 12 infected chambers had no growth of C. pseudotuberculosis 15 days postadministration. Results of this study indicate that tulathromycin may be beneficial in the treatment of focal infections such as those caused by C. pseudotuberculosis.
Figures


Similar articles
-
Oxytetracycline concentrations in interstitial fluid from tissue chambers inoculated with Corynebacterium pseudotuberculosis after intramuscular or intrachamber administration in sheep.Am J Vet Res. 2019 Jun;80(6):586-594. doi: 10.2460/ajvr.80.6.586. Am J Vet Res. 2019. PMID: 31140853
-
Tulathromycin assay validation and tissue residues after single and multiple subcutaneous injections in domestic goats (Capra aegagrus hircus).J Vet Pharmacol Ther. 2012 Apr;35(2):113-20. doi: 10.1111/j.1365-2885.2011.01300.x. Epub 2011 Jul 18. J Vet Pharmacol Ther. 2012. PMID: 21762403 Clinical Trial.
-
Pharmacokinetics and tissue elimination of tulathromycin following subcutaneous administration in meat goats.Am J Vet Res. 2012 Oct;73(10):1634-40. doi: 10.2460/ajvr.73.10.1634. Am J Vet Res. 2012. PMID: 23013191
-
Effects of experimentally induced respiratory disease on the pharmacokinetics and tissue residues of tulathromycin in meat goats.J Vet Pharmacol Ther. 2019 Jul;42(4):420-429. doi: 10.1111/jvp.12764. Epub 2019 Jun 10. J Vet Pharmacol Ther. 2019. PMID: 31183876
-
Tulathromycin: an overview of a new triamilide antibiotic for livestock respiratory disease.Vet Ther. 2005 Summer;6(2):83-95. Vet Ther. 2005. PMID: 16094557 Review.
Cited by
-
Gross and Histopathology of Goats Feeding on Opuntia stricta in Laikipia County, Kenya.Vet Med Int. 2021 Feb 3;2021:8831996. doi: 10.1155/2021/8831996. eCollection 2021. Vet Med Int. 2021. PMID: 33623658 Free PMC article.
-
Brazilian Green Propolis as a Therapeutic Agent for the Post-surgical Treatment of Caseous Lymphadenitis in Sheep.Front Vet Sci. 2019 Nov 26;6:399. doi: 10.3389/fvets.2019.00399. eCollection 2019. Front Vet Sci. 2019. PMID: 31850377 Free PMC article.
References
-
- Mills AE, Mitchell RD, Lim EK. 1997. Corynebacterium pseudotuberculosis is a cause of human necrotising granulomatous lymphadenitis. Pathology 29:231–233 - PubMed
-
- Peel MM, Palmer GG, Stacpoole AM, Kerr TG. 1997. Human lymphadenitis due to Corynebacterium pseudotuberculosis: report of ten cases from Australia and review. Clin. Infect. Dis. 24:185–191 - PubMed
-
- Williamson LH. 2001. Caseous lymphadenitis in small ruminants. Vet. Clin. North Am. Food Anim. Pract. 17:359–371 - PubMed
-
- Rizvi S, Green LE, Glover MJ. 1997. Caseous lymphadenitis: an increasing cause for concern. Vet. Rec. 140(22):586–587 - PubMed
-
- Stoops SG, Renshaw HW, Thilsted JP. 1984. Ovine caseous lymphadenitis: disease prevalence, lesion distribution, and thoracic manifestations in a population of mature culled sheep from western United States. Am. J. Vet. Res. 45:557–561 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

