Correlates of immune protection following cutaneous immunization with an attenuated Burkholderia pseudomallei vaccine
- PMID: 24101688
- PMCID: PMC3837973
- DOI: 10.1128/IAI.00915-13
Correlates of immune protection following cutaneous immunization with an attenuated Burkholderia pseudomallei vaccine
Abstract
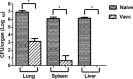
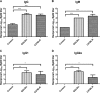
Infections with the Gram-negative bacterium Burkholderia pseudomallei (melioidosis) are associated with high mortality, and there is currently no approved vaccine to prevent the development of melioidosis in humans. Infected patients also do not develop protective immunity to reinfection, and some individuals will develop chronic, subclinical infections with B. pseudomallei. At present, our understanding of what constitutes effective protective immunity against B. pseudomallei infection remains incomplete. Therefore, we conducted a study to elucidate immune correlates of vaccine-induced protective immunity against acute B. pseudomallei infection. BALB/c and C57BL/6 mice were immunized subcutaneously with a highly attenuated, Select Agent-excluded purM deletion mutant of B. pseudomallei (strain Bp82) and then subjected to intranasal challenge with virulent B. pseudomallei strain 1026b. Immunization with Bp82 generated significant protection from challenge with B. pseudomallei, and protection was associated with a significant reduction in bacterial burden in lungs, liver, and spleen of immunized mice. Humoral immunity was critically important for vaccine-induced protection, as mice lacking B cells were not protected by immunization and serum from Bp82-vaccinated mice could transfer partial protection to nonvaccinated animals. In contrast, vaccine-induced protective immunity was found to be independent of both CD4 and CD8 T cells. Tracking studies demonstrated uptake of the Bp82 vaccine strain predominately by neutrophils in vaccine-draining lymph nodes and by smaller numbers of dendritic cells (DC) and monocytes. We concluded that protection following cutaneous immunization with a live attenuated Burkholderia vaccine strain was dependent primarily on generation of effective humoral immune responses.
Figures


Similar articles
-
Evaluation of Burkholderia mallei ΔtonB Δhcp1 (CLH001) as a live attenuated vaccine in murine models of glanders and melioidosis.PLoS Negl Trop Dis. 2019 Jul 15;13(7):e0007578. doi: 10.1371/journal.pntd.0007578. eCollection 2019 Jul. PLoS Negl Trop Dis. 2019. PMID: 31306423 Free PMC article.
-
Burkholderia pseudomallei ΔtonB Δhcp1 Live Attenuated Vaccine Strain Elicits Full Protective Immunity against Aerosolized Melioidosis Infection.mSphere. 2019 Jan 2;4(1):e00570-18. doi: 10.1128/mSphere.00570-18. mSphere. 2019. PMID: 30602524 Free PMC article.
-
Novel multi-component vaccine approaches for Burkholderia pseudomallei.Clin Exp Immunol. 2019 May;196(2):178-188. doi: 10.1111/cei.13286. Epub 2019 Apr 8. Clin Exp Immunol. 2019. PMID: 30963550 Free PMC article. Review.
-
Multicomponent Gold-Linked Glycoconjugate Vaccine Elicits Antigen-Specific Humoral and Mixed TH1-TH17 Immunity, Correlated with Increased Protection against Burkholderia pseudomallei.mBio. 2021 Jun 29;12(3):e0122721. doi: 10.1128/mBio.01227-21. Epub 2021 Jun 29. mBio. 2021. PMID: 34182777 Free PMC article.
-
Development of Burkholderia mallei and pseudomallei vaccines.Front Cell Infect Microbiol. 2013 Mar 11;3:10. doi: 10.3389/fcimb.2013.00010. eCollection 2013. Front Cell Infect Microbiol. 2013. PMID: 23508691 Free PMC article. Review.
Cited by
-
Recent Advances in Burkholderia mallei and B. pseudomallei Research.Curr Trop Med Rep. 2015 Jun;2(2):62-69. doi: 10.1007/s40475-015-0042-2. Curr Trop Med Rep. 2015. PMID: 25932379 Free PMC article.
-
Functional Activities of O-Polysaccharide and Hemolysin Coregulated Protein 1 Specific Antibodies Isolated from Melioidosis Patients.Infect Immun. 2022 Nov 17;90(11):e0021422. doi: 10.1128/iai.00214-22. Epub 2022 Oct 13. Infect Immun. 2022. PMID: 36226942 Free PMC article.
-
Evaluation of Burkholderia mallei ΔtonB Δhcp1 (CLH001) as a live attenuated vaccine in murine models of glanders and melioidosis.PLoS Negl Trop Dis. 2019 Jul 15;13(7):e0007578. doi: 10.1371/journal.pntd.0007578. eCollection 2019 Jul. PLoS Negl Trop Dis. 2019. PMID: 31306423 Free PMC article.
-
Burkholderia pseudomallei ΔtonB Δhcp1 Live Attenuated Vaccine Strain Elicits Full Protective Immunity against Aerosolized Melioidosis Infection.mSphere. 2019 Jan 2;4(1):e00570-18. doi: 10.1128/mSphere.00570-18. mSphere. 2019. PMID: 30602524 Free PMC article.
-
Novel multi-component vaccine approaches for Burkholderia pseudomallei.Clin Exp Immunol. 2019 May;196(2):178-188. doi: 10.1111/cei.13286. Epub 2019 Apr 8. Clin Exp Immunol. 2019. PMID: 30963550 Free PMC article. Review.
References
-
- Wiersinga WJ, van der Poll T, White NJ, Day NP, Peacock SJ. 2006. Melioidosis: insights into the pathogenicity of Burkholderia pseudomallei. Nat. Rev. Microbiol. 4:272–282 - PubMed
-
- Wiersinga WJ, Currie BJ, Peacock SJ. 2012. Melioidosis. N. Engl. J. Med. 367:1035–1044 - PubMed
-
- Dance DAB. 2000. Melioidosis as an emerging global problem. Acta Trop. 74:115–119 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

