Dissecting functions of the conserved oligomeric Golgi tethering complex using a cell-free assay
- PMID: 24102787
- PMCID: PMC3892563
- DOI: 10.1111/tra.12128
Dissecting functions of the conserved oligomeric Golgi tethering complex using a cell-free assay
Abstract
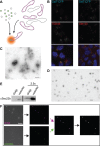
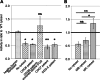
Vesicle transport sorts proteins between compartments and is thereby responsible for generating the non-uniform protein distribution along the eukaryotic secretory and endocytic pathways. The mechanistic details of specific vesicle targeting are not yet well characterized at the molecular level. We have developed a cell-free assay that reconstitutes vesicle targeting utilizing the recycling of resident enzymes within the Golgi apparatus. The assay has physiological properties, and could be used to show that the two lobes of the conserved oligomeric Golgi tethering complex play antagonistic roles in trans-Golgi vesicle targeting. Moreover, we can show that the assay is sensitive to several different congenital defects that disrupt Golgi function and therefore cause glycosylation disorders. Consequently, this assay will allow mechanistic insight into the targeting step of vesicle transport at the Golgi, and could also be useful for characterizing some novel cases of congenital glycosylation disorders.
Keywords: Golgi apparatus; cell-free reconstitution; congenital disorders of glycosylation; conserved oligomeric Golgi complex; glycosylation enzyme sorting; vesicle tethering.
©2013 The Authors. Traffic published by John Wiley & Sons Ltd.
Figures

References
-
- Barlowe C, Orci L, Yeung T, Hosobuchi M, Hamamoto S, Salama N, Rexach MF, Ravazzola M, Amherdt M, Schekman R. Copii – a membrane coat formed by sec proteins that drive vesicle budding from the endoplasmic-reticulum. Cell. 1994;77:895–907. - PubMed
-
- Weber T, Zemelman BV, McNew JA, Westermann B, Gmachl M, Parlati F, Sollner TH, Rothman JE. SNAREpins: minimal machinery for membrane fusion. Cell. 1998;92:759–772. - PubMed
-
- Balch WE, Dunphy WG, Braell WA, Rothman JE. Reconstitution of the transport of protein between successive compartments of the Golgi measured by the coupled incorporation of N-acetylglucosamine. Cell. 1984;39:405–416. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

