Ozone induces glucose intolerance and systemic metabolic effects in young and aged Brown Norway rats
- PMID: 24103449
- PMCID: PMC4343260
- DOI: 10.1016/j.taap.2013.09.029
Ozone induces glucose intolerance and systemic metabolic effects in young and aged Brown Norway rats
Abstract
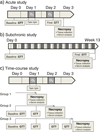
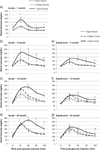
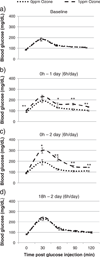
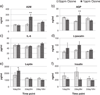
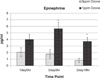
Air pollutants have been associated with increased diabetes in humans. We hypothesized that ozone would impair glucose homeostasis by altering insulin signaling and/or endoplasmic reticular (ER) stress in young and aged rats. One, 4, 12, and 24 month old Brown Norway (BN) rats were exposed to air or ozone, 0.25 or 1.0 ppm, 6 h/day for 2 days (acute) or 2 d/week for 13 weeks (subchronic). Additionally, 4 month old rats were exposed to air or 1.0 ppm ozone, 6 h/day for 1 or 2 days (time-course). Glucose tolerance tests (GTT) were performed immediately after exposure. Serum and tissue biomarkers were analyzed 18 h after final ozone for acute and subchronic studies, and immediately after each day of exposure in the time-course study. Age-related glucose intolerance and increases in metabolic biomarkers were apparent at baseline. Acute ozone caused hyperglycemia and glucose intolerance in rats of all ages. Ozone-induced glucose intolerance was reduced in rats exposed for 13 weeks. Acute, but not subchronic ozone increased α2-macroglobulin, adiponectin and osteopontin. Time-course analysis indicated glucose intolerance at days 1 and 2 (2>1), and a recovery 18 h post ozone. Leptin increased day 1 and epinephrine at all times after ozone. Ozone tended to decrease phosphorylated insulin receptor substrate-1 in liver and adipose tissues. ER stress appeared to be the consequence of ozone induced acute metabolic impairment since transcriptional markers of ER stress increased only after 2 days of ozone. In conclusion, acute ozone exposure induces marked systemic metabolic impairments in BN rats of all ages, likely through sympathetic stimulation.
Keywords: Aging; Air pollution; Epinephrine; Metabolic syndrome; Ozone; Serum biomarkers.
© 2013.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures

References
-
- Adar SD, Sheppard L, Vedal S, Polak JF, Sampson PD, Diez Roux AV, Budoff M, Jacobs DR, Jr, Barr RG, Watson K, Kaufman JD. Fine particulate air pollution and the progression of carotid intima-medial thickness: a prospective cohort study from the multi-ethnic study of atherosclerosis and air pollution. PLoS Med. 2013;10:e1001430. - PMC - PubMed
-
- Blackburn WD., Jr Validity of acute phase proteins as markers of disease activity. J. Rheumatol. Suppl. 1994;42:9–13. - PubMed
-
- Brennan AM, Mantzoros CS. Drug insight: the role of leptin in human physiology and pathophysiology—emerging clinical applications. Nat. Clin. Pract. Endocrinol. Metab. 2006;2:318–327. - PubMed
-
- Brook RD, Rajagopalan S. Particulate matter air pollution and atherosclerosis. Curr. Atheroscler. Rep. 2010;12:291–300. - PubMed
-
- Brook RDMD, Jerrett MP, Brook JRP, Bard RLMA, Finkelstein MMMDP. The relationship between diabetes mellitus and traffic-related air pollution. J. Occup. Environ. Med. 2008;50:32–38. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

