Histone demethylase Jmjd3 regulates osteoblast differentiation via transcription factors Runx2 and osterix
- PMID: 24106268
- PMCID: PMC3837102
- DOI: 10.1074/jbc.M113.497040
Histone demethylase Jmjd3 regulates osteoblast differentiation via transcription factors Runx2 and osterix
Abstract
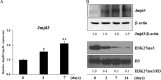
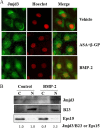
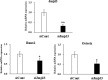
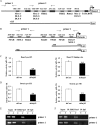
Post-translational modifications of histones including methylation play important roles in cell differentiation. Jumonji domain-containing 3 (Jmjd3) is a histone demethylase, which specifically catalyzes the removal of trimethylation of histone H3 at lysine 27 (H3K27me3). In this study, we examined the expression of Jmjd3 in osteoblasts and its roles in osteoblast differentiation. Jmjd3 expression in the nucleus was induced in response to the stimulation of osteoblast differentiation as well as treatment of bone morphogenetic protein-2 (BMP-2). Either treatment with Noggin, an inhibitor of BMP-2, or silencing of Smad1/5 suppressed Jmjd3 expression during osteoblast differentiation. Silencing of Jmjd3 expression suppressed osteoblast differentiation through the expression of bone-related genes including Runx2, osterix, osteopontin, bone sialoprotein (BSP), and osteocalcin (OCN). Silencing of Jmjd3 decreased the promoter activities of Runx2 and osterix and increased the level of H3K27me3 on the promoter regions of Runx2 and osterix. Introduction of the exogenous Runx2 and osterix partly rescued osteoblast differentiation in the shJmjd3 cells. The present results indicate that Jmjd3 plays important roles in osteoblast differentiation and regulates the expressions of BSP and OCN via transcription factors Runx2 and osterix.
Keywords: Bone; Bone morphogenetic protein (BMP); Differentiation; Histone methylation; Osteoblasts.
Figures





Similar articles
-
Histone demethylase Utx regulates differentiation and mineralization in osteoblasts.J Cell Biochem. 2015 Nov;116(11):2628-36. doi: 10.1002/jcb.25210. J Cell Biochem. 2015. PMID: 25920016
-
Histone demethylase JMJD3 is required for osteoblast differentiation in mice.Sci Rep. 2015 Aug 25;5:13418. doi: 10.1038/srep13418. Sci Rep. 2015. PMID: 26302868 Free PMC article.
-
Histone demethylase Jmjd3 modulates osteoblast apoptosis induced by tumor necrosis factor-alpha through directly targeting RASSF5.Connect Tissue Res. 2020 Nov;61(6):517-525. doi: 10.1080/03008207.2019.1620225. Epub 2019 May 27. Connect Tissue Res. 2020. PMID: 31092054
-
The Roles of Histone Demethylase Jmjd3 in Osteoblast Differentiation and Apoptosis.J Clin Med. 2017 Feb 23;6(3):24. doi: 10.3390/jcm6030024. J Clin Med. 2017. PMID: 28241471 Free PMC article. Review.
-
Emerging roles of JMJD3 in cancer.Clin Transl Oncol. 2022 Jul;24(7):1238-1249. doi: 10.1007/s12094-021-02773-9. Epub 2022 Mar 3. Clin Transl Oncol. 2022. PMID: 35239138 Review.
Cited by
-
Cellular behaviours of bone marrow-derived mesenchymal stem cells towards pristine graphene oxide nanosheets.Cell Prolif. 2017 Oct;50(5):e12367. doi: 10.1111/cpr.12367. Epub 2017 Aug 3. Cell Prolif. 2017. PMID: 28771866 Free PMC article.
-
Tet-Mediated DNA Demethylation Is Required for SWI/SNF-Dependent Chromatin Remodeling and Histone-Modifying Activities That Trigger Expression of the Sp7 Osteoblast Master Gene during Mesenchymal Lineage Commitment.Mol Cell Biol. 2017 Sep 26;37(20):e00177-17. doi: 10.1128/MCB.00177-17. Print 2017 Oct 15. Mol Cell Biol. 2017. PMID: 28784721 Free PMC article.
-
The inhibition of EZH2 ameliorates osteoarthritis development through the Wnt/β-catenin pathway.Sci Rep. 2016 Aug 19;6:29176. doi: 10.1038/srep29176. Sci Rep. 2016. PMID: 27539752 Free PMC article.
-
Icariin promotes osteogenic differentiation through the mmu_circ_0000349/mmu-miR-138-5p/Jumonji domain-containing protein-3 axis.Heliyon. 2023 Nov 6;9(11):e21885. doi: 10.1016/j.heliyon.2023.e21885. eCollection 2023 Nov. Heliyon. 2023. PMID: 38045146 Free PMC article.
-
Osteogenic and Chondrogenic Potential of Periosteum-Derived Mesenchymal Stromal Cells: Do They Hold the Key to the Future?Pharmaceuticals (Basel). 2021 Nov 8;14(11):1133. doi: 10.3390/ph14111133. Pharmaceuticals (Basel). 2021. PMID: 34832915 Free PMC article. Review.
References
-
- Chen D., Harris M. A., Rossini G., Dunstan C. R., Dallas S. L., Feng J. Q., Mundy G. R., Harris S. E. (1997) Bone morphogenetic protein 2 (BMP-2) enhances BMP-3, BMP-4, and bone cell differentiation marker gene expression during the induction of mineralized bone matrix formation in cultures of fetal rat calvarial osteoblasts. Calcif. Tissue Int. 60, 283–290 - PubMed
-
- Wozney J. M., Rosen V. (1998) Bone morphogenetic protein and bone morphogenetic protein gene family in bone formation and repair. Clin. Orthop. Relat. Res. 26–37 - PubMed
-
- Hanai J., Chen L. F., Kanno T., Ohtani-Fujita N., Kim W. Y., Guo W. H., Imamura T., Ishidou Y., Fukuchi M., Shi M. J., Stavnezer J., Kawabata M., Miyazono K., Ito Y. (1999) Interaction and functional cooperation of PEBP2/CBF with Smads: synergistic induction of the immunoglobulin germline Cα promoter. J. Biol. Chem. 274, 31577–31582 - PubMed
-
- Ducy P., Zhang R., Geoffroy V., Ridall A. L., Karsenty G. (1997) Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell 89, 747–754 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

